By Charlotte Vallaeys
 Some of my favorite memories of my sons as babies (which was not that long ago—they are 2 and 4 years old) involve breastfeeding. It is a truly magical and priceless bonding experience. And for someone who loves good, “real” food as much as I do, there could be nothing more satisfying and empowering than that tingling feeling that accompanies the milk ducts springing into action to produce fresh milk, full of just the right mix of enzymes, nutrients, antibodies, hormones, and other beneficial components that have yet to be discovered—the product of millions of years of evolution, specially made for my baby to ensure he’ll grow and thrive.
Some of my favorite memories of my sons as babies (which was not that long ago—they are 2 and 4 years old) involve breastfeeding. It is a truly magical and priceless bonding experience. And for someone who loves good, “real” food as much as I do, there could be nothing more satisfying and empowering than that tingling feeling that accompanies the milk ducts springing into action to produce fresh milk, full of just the right mix of enzymes, nutrients, antibodies, hormones, and other beneficial components that have yet to be discovered—the product of millions of years of evolution, specially made for my baby to ensure he’ll grow and thrive.
But we also faced many obstacles along the way. The challenges started in the hospital with my firstborn and continued up to the moment my lastborn sipped his last drop of human milk.
My oldest, Liam, would not latch on at first. I had expected breastfeeding to be easy—it was, after all, so “natural”—but there I was, a brand new mom with an hours-old baby, struggling to get my newborn to latch on. Those first hours of motherhood ushered in the realization that, in parenthood, not everything will go as planned.
After involving many contraptions, including a hospital-grade breast pump, specialized bottles, and a silicone “nipple shield,” and many different nurses’ and lactation consultants’ advice (not to mention a lot of maternal determination), we finally—after a couple of days—made it work.
When Liam was 9 weeks old, I discovered blood in his diaper and disregarded our pediatrician’s advice to switch from breastfeeding to hypoallergenic formula (she claimed he was allergic to milk, including his mother’s milk). Instead, I continued breastfeeding but cut all dairy and soy out of my diet until Liam’s first birthday.
Among other challenges, we weathered two bouts of painful mastitis and many days of separation for work-related trips that required a freezer full of pumped milk.
The challenges continued until the very end, and unfortunately, my final memories of breastfeeding are also the most painful. When he was 9 months, my second son, Kai, decided to wean on his own. I had heard about “self-weaning,” and until I was faced with an uninterested and stubborn infant, I thought it was just a clever excuse for mothers to switch to more convenient formula feeding. Again, it was a stark reminder that many things about parenthood are easier said than done, and so much of what happens on this journey is entirely out of our control.
I suppose I was more stubborn than Kai, and he eventually became hungry and thirsty enough that he would feed. But he responded by reluctantly drinking and then ending the session with a bite. After a couple of weeks of very tense—and often painful—feeding sessions, I switched to pumping and giving my milk to Kai from a bottle. I grimaced every time I saw him bite down on that rubber nipple. Eventually, after a couple more weeks, I produced no more milk, and that bottle needed something in it for the remaining weeks until we could switch to organic whole milk.
All this baby-feeding drama happened while I worked as a researcher and policy analyst with The Cornucopia Institute. Cornucopia acts as a watchdog for the organic community. As I carefully read labels in the baby food aisle of my local food store, I couldn’t help but notice numerous violations of the organic standards. I avoided these baby foods with multiple unapproved synthetic ingredients. Meanwhile, Cornucopia took a leading role in advocating for the removal of unnecessary or potentially harmful synthetics from organic formula and baby food.
This post is for parents, grandparents and others who want more information on organic infant formula. I’d like to share what I learned both as Policy Director at Cornucopia and as a mom looking for the best food for my own babies.
Without a doubt, human milk and factory-produced infant formula don’t compare, as human milk is far superior in so many respects, including in ways we will probably never fully comprehend. We can all agree on that. But in parenthood, many things don’t go as planned, and for many committed, food-conscious, organic-buying parents, that includes breastfeeding.
I will only cover organic formula, and I hope that readers will understand that while there are many problems in this segment of the organic industry, organic formula is still a far better choice than conventional formula, with its genetically engineered ingredients (GMOs), milk from cows that were likely treated with antibiotics or artificial growth hormones, and oils that were processed with the use of neurotoxic solvents like hexane. Major ingredients in conventional formula are derived from crops that were sprayed with harmful pesticides and herbicides in the field and likely fumigated in storage.
Organic formula offers an alternative, but it is far from perfect. I hope that parents will find the information here useful if faced with the tough decision to turn to formula.
The Organic Formula Industry
If you need formula and can’t make your own, you likely want to know how to purchase the best one.
While there are seven brands of organic infant formula currently available on market shelves, there are only three companies that make organic infant formula in the United States.
One manufacturer is PBM Nutritionals, owned by Perrigo. At $3 billion in annual sales, Perrigo is the world’s largest manufacturer of private label (store-brand) over-the-counter pharmaceuticals. PBM primarily produces conventional formula, but makes organic formula for its own Vermont Organics and Bright Beginnings brands. It also manufacturers organic formula for Hain Celestial’s Earth’s Best brand, Whole Foods Market’s 365 Organic brand, and Walmart’s Parent’s Choice brand.
Similac Organic is manufactured by Abbott Laboratories. Abbott, a major manufacturer of pharmaceuticals in the U.S., introduced Similac Organic in 2006. By 2007, its first full year on sale, Similac Organic captured 36% of the organic formula market. Abbott’s Similac is a market leader in conventional formula.
Finally, Baby’s Only Organic is developed and marketed by Nature’s One. Nature’s One markets Baby’s Only Organic formula as a “toddler formula” rather than an infant formula (according to the company, this is done to encourage breastfeeding until age 1). Its products meet the same nutritional standards that the FDA sets forth for infant formula. Nature’s One is the only company marketing organic formula that is not a publicly traded corporation; the business is family-owned and operated.
Top five ingredients to take a close look at – how do organic brands compare?
1. Sweeteners: corn syrup, sugar, or brown rice syrup
Formula manufacturers strive to formulate a product that mirrors the nutritional profile of human milk. Human milk contains higher levels of lactose, a carbohydrate, than cow milk, which means that formula manufacturers must make up the difference by adding a sweetener to cow milk-based formula.
But in their choice of sweetener, it appears that concerns over the availability and price of the various sources have taken precedence. The sweetener that most closely mimics human milk would be lactose (the naturally occurring carbohydrate in any mammal’s milk). But lactose is also the most expensive, and manufacturers have, over the years, switched from this milk-based sweetener to plant-based sweeteners.
Corn syrup
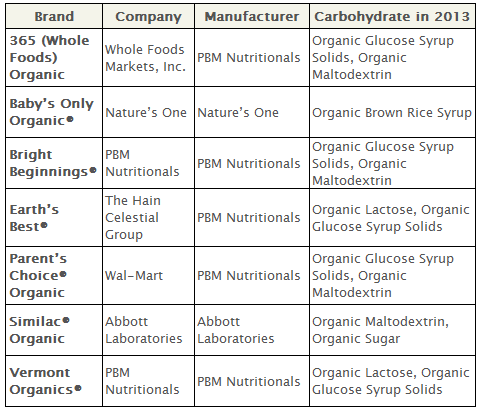
When PBM Nutritionals first rolled out its organic infant formula under the Bright Beginnings brand name, it contained only organic lactose, with no corn-based sweeteners. PBM soon produced the same product for Walmart, under the Parent’s Choice brand name, which also contained only organic lactose.
But PBM Nutritionals switched from organic lactose as the sole sweetener for Bright Beginnings, and so did Walmart’s organic formula, in 2010. Their formulations changed to include both ‘organic corn syrup solids’ and lactose.
By 2011, organic lactose in Bright Beginnings and Parent’s Choice had disappeared altogether, replaced by organic maltodextrin, another plant-based sweetener. Maltodextrins are partially hydrolyzed starch molecules, which can be derived from corn, rice or potatoes. Maltodextrin is less sweet than corn syrup.
Today, Bright Beginnings, Parent’s Choice and Whole Foods’ 365 Organic contain no organic lactose at all—only plant-based (mostly corn-based) carbohydrates.
A similar move away from organic lactose happened with Earth’s Best and Vermont Organics (again, both manufactured by PBM). In 2007, Earth’s Best infant formula contained only organic lactose as the added carbohydrate. When Vermont Organics entered the market in 2008, it mirrored Earth’s Best and also contained only organic lactose. By 2011, both Earth’s Best and Vermont Organics contained reduced amounts of organic lactose, which were replaced with ‘organic glucose syrup solids.’
‘Organic glucose syrup solids’ is another name for ‘organic corn syrup solids,’ which are partially hydrolyzed corn starch molecules that are dried to a low moisture powder (hence the name ‘solids’). Corn syrup solids are moderately sweet (sweeter than maltodextrin).
Sugar
In terms of the added sweetener, Abbott Laboratories took a different route for its Similac Organic product: sugar.
Abbott’s decision came under fire in 2008, when The New York Times reported on the various concerns regarding the use of sugar in infant formula. The Times even commissioned its own professional taste test. Similac Organic was the sweetest, “with the sweetness of grape juice or Country Time lemonade,” according to Gail Civille, the director of Sensory Spectrum, which performed the tests.
The European Union banned sugar-sweetened infant formula in 2009, due to concerns with rising rates of childhood obesity and the possibility that overly sweet formula might lead to overfeeding. Sucrose (sugar) is allowed only in special formula for babies with allergies, and even then, it may not exceed 20% of the total carbohydrate content.
The New York Times quoted Dr. Benjamin Caballero, director of the Center for Human Nutrition at the Johns Hopkins Bloomberg School of Public Health and an expert in risk factors for childhood obesity: “I would be very concerned about this as a pediatrician. The issue is that sweet tastes tend to encourage consumption of excessive amounts.” Evidence shows that babies and children will always show a preference for the sweetest food available, he said, and they will eat more of it than they would of less-sweet food. “This is how breakfast cereal manufacturers compete,” he added.
Despite the concerns raised in The New York Times, Abbott Laboratories continues to use sugar as the added carbohydrate.
Brown Rice Syrup
Nature’s One chose organic brown rice syrup as the added carbohydrate for its toddler formula. In 2012, researchers at Dartmouth University tested various foods for levels of arsenic, and found organic toddler formula made with organic brown rice syrup contained up to six times the U.S. EPA safe drinking water limit for inorganic arsenic (there are no established safety standards for arsenic in food, including infant formula).
In response, Nature’s One developed an organic-compliant technology to filter and remove inorganic arsenic from its organic brown rice syrup to undetectable levels.
Consumers Union tested Baby’s Only Organic products to determine if the company had indeed removed arsenic from its products, and confirmed Nature’s One’s claims that its formula now contains undetectable arsenic levels
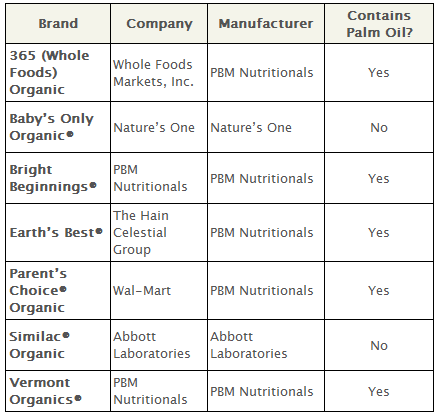
2. Palm Oil: Forms “Soaps” In the Baby’s Gut
Not all oil is created equal—it’s a basic fact of nutrition science, and one that is especially important for infants. Human milk is naturally high in certain types of fatty acids, which formula manufacturers try to mimic by adding certain types of oil. And to mirror the levels of palmitic acid, a fatty acid found in human milk, some manufacturers add palm oil.
However, palmitic acid from palm oil is structurally different from palmitic acid in human milk, and research has shown that human infants do not properly absorb it.
The unabsorbed palmitic acid remaining in the infant’s gut reacts with calcium, and causes the formation of “soaps” in the baby’s intestines. This important finding has been reported on more than one occasion in the journa lPediatrics, of the American Academy of Pediatrics.
Research suggests that the formation of “soaps” in the baby’s intestinal tract negatively affects a baby’s development and health. Since palmitic acid from palm oil is not absorbed properly, it means overall fat absorption is lower in babies given formula with palm oil. Bone mass is significantly lower in babies given formula containing palm oil, perhaps because the calcium in the baby’s intestines turns into “soap” rather than reaching the baby’s growing bones. And the “soapiness” in the intestines also leads to hard stools.
According to researchers at Wayne State University, who performed a comprehensive review of published studies on the effects of palm oil in infant formula:
The use of palm oil in infant formulas to match the human milk content of palmitic acid has unintended physiological consequences. The avoidance of palm oil in infant formulas can prevent this detrimental effect.
Despite these concerns, all organic formula products coming out of the PBM Nutritionals factory continue to contain palm oil. Earth’s Best did not contain palm oil when it first came on the market, but switched to palm oil in 2007.
(As if parents needed another reason to avoid Enfamil, which offers no organic option, the first oil listed in Enfamil is palm oil).
3. Ingredients extracted with neurotoxic solvents from algae and soil fungus—not exactly the way mom makes it – C. Cohnii oil (DHA) and M. Alpina oil (ARA):
Six years ago, Cornucopia’s codirectors asked me to look into two ingredients, C. Cohnii oil and M. Alpina oil. They are marketed as “DHA and ARA,” and were starting to appear in organic infant formula without having gone through the proper approval process required by federal organic law.
The oils were manufactured by a biotechnology corporation in Maryland (the company has since been bought by the Dutch multinational corporation Royal DSM), using processing aids and synthetic ingredients that are not approved for use in organics.
Fresh out of a graduate program in nutrition, I could not help but dig deeper. I have to admit: I didn’t want to be involved in filing a legal complaint against ingredients that might be beneficial to infant development. I was concerned we might be depriving infants if we actually succeeded in having the USDA pull these ingredients out of organic infant formula (I clearly underestimated the power of the infant formula lobby that we’d be up against).
I soon discovered from the scientific literature that we wouldn’t be harming babies at all if they didn’t have these additives in their formula. If fact, we’d be protecting them from potential harm. Studies repeatedly failed to show benefits from adding these additives to formula, and the FDA had been receiving dozens of reports from parents and pediatricians who noticed some babies do not tolerate these ingredients. In 2008, we filed a legal complaint.
DHA is an omega-3 fatty acid, and ARA is an omega-6 fatty acid. Both are naturally found in human milk, and DHA is a component of brain and eye tissue. But the DHA and ARA added to most brands of infant formula are extracted from factory-produced C. Cohnii and M. Alpina—specific strains of algae and fungus that have never been part of the human diet, let alone the diet of infants.
Since we released our report and filed the legal complaint, it has become even clearer that these additives are not necessary and are primarily added as marketing tools. Three of the most prominent and respected independent scientists in the field of infant formula science stated in 2010 that the scientific evidence base for DHA and ARA’s addition to infant formula is “recognized by most investigators and Key Opinion Leaders in the field to be weak,” and that “this field of research has been driven to an extent by enthusiasm and vested interest.”
Several comprehensive reviews of all published research have been conducted since we released our report, and all conclude that DHA and ARA “had no proven benefit regarding vision, cognition, or physical growth.” The World Health Organization’s Director of Nutrition for Health and Development even wrote a letter in 2011 to members of the European parliament, letting them know that “as to date no solid evidence exists to be able to say that adding DHA to infant formula will have important clinical benefits.”
When an Associated Press reporter asked the chair of the American Academy of Pediatrics committee on nutrition, Dr. Frank Greer, to comment on DHA and ARA in infant formula, he said: “The truth of the matter is, they’re not essential. Humans can synthesize these. Fatty acids are naturally present in the diet. And the whole issue becomes, do you make really make people smarter if you put DHA and ARA in everything? Or is this just all marketing hype? Personally, I lean toward the latter.”
When the C. Cohnii and M. Alpina oils first appeared in infant formula, the Food and Drug Administration (FDA) received dozens of reports from physicians and parents who noticed diarrhea, vomiting, and other gastrointestinal distress in infants given formula with these oils—symptoms that disappeared when the infant was switched to the exact same formula without these novel additives.
The FDA reports that no post-market surveillance has taken place to assure the safety and tolerance of these additives—despite the FDA’s clear request to the formula manufacturers to conduct rigorous post-market monitoring to ensure their safety.
Another concern with these additives is how they are produced: the oil is extracted from the algae and fungus with the use of hexane, a neurotoxic petroleum-based solvent.
When the USDA received legal complaints against the unapproved use of C. Cohnii and M. Alpina oils, a Washington lobbyist with the powerful law firm of Covington and Burling convinced USDA officials to open a loophole in the organic standards, which would allow the formula manufacturers to add the unapproved ingredients without facing enforcement action.
While the USDA has since admitted that this was inappropriate, the agency has failed to take enforcement action and continues to bow to pressure from the infant formula industry. The National Organic Standards Board even explicitly stated that hexane-extracted algal oil and fungal oil should not be allowed in organic foods— but the USDA has failed to act on this very clear and legally binding vote, and hexane-extracted DHA and ARA remains in organic infant formula.
The only company that adheres to the legal requirement that DHA and ARA oil must not be extracted with the use of the neurotoxic solvent hexane is Nature’s One, which has chosen a water-extracted source of DHA and ARA (derived from egg yolks) for its Baby’s Only Organic formula.
4. Carrageenan: Dangerous Inflammation In Your Baby’s Gut
If you search for “carrageenan” in a medical database, thousands of search results will appear. Why so many? Because carrageenan is used in animal experiments to predictably cause inflammation, which allows pharmaceutical scientists to test the effectiveness of new anti-inflammatory drugs.
In a report Cornucopia released earlier this year, we carefully analyzed the scientific literature on food-grade carrageenan, and found that scientists have raised concern about carrageenan’s safety for decades. These concerns are based on their research linking the common food additive to gastrointestinal disease in laboratory animals, including colon tumors.
But the food industry, including the infant formula industry, has responded for decades by claiming that carrageenan is safe—based largely on industry-funded studies, with flawed methodologies. When a Chicago Tribune reporter asked both the FDA and the carrageenan industry lobby group earlier this year to share studies that were not funded by the industry and that could indicate carrageenan is safe, they could not come up with a single one.
Carrageenan appears in some organic infant formula, even though the National Organic Standards Board (NOSB) voted to prohibit it. The Secretary of Agriculture’s decision to disregard the NOSB’s decision shows the lobbying power and influence of the infant formula industry.
Carrageenan is prohibited in infant formula—conventional and organic—in the European Union. The science linking carrageenan to intestinal inflammation is disturbing enough, but what adds insult to injury is that it is entirely unnecessary. Carrageenan contributes no nutritional value or flavor to formula, or other food, but is added to stabilize ready-to-feed formula. Adding carrageenan means parents or caregivers do not have to shake the product before feeding it to the baby. The alternative is to put a “shake well” label on the bottle.
Earth’s Best and Similac Organic ready-to-feed formula, the only liquid organic formula on the market, both contain carrageenan.
5. Synthetic Preservatives and Nutrients In Organics: A Mirror-Image Of The Synthetics In Conventional Formula
Federal law requires that a synthetic ingredient cannot be added to organic products unless it has been reviewed and approved by the National Organic Standards Board (NOSB). Two synthetic preservatives and numerous synthetic nutrients have recently been rejected by the NOSB. Final rulemaking is pending. For now, they remain in some organic infant formula products.
Some brands contain more of these unapproved synthetics than others.
Synthetic Preservatives
The NOSB rejected two synthetic preservatives: ascorbyl palmitate and beta-carotene. One of the primary reasons why formula manufacturers add these synthetic preservatives is to prevent the algal DHA and fungal ARA oils from going rancid. Since Baby’s Only Organic is the only formula that does not contain algal DHA oil and fungal ARA oil, it also is the only formula that does not contain these two synthetic preservatives.
Synthetic Nutrients
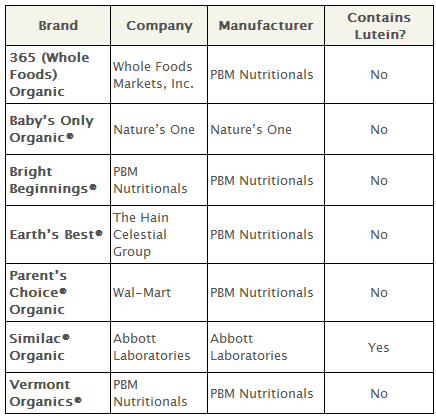
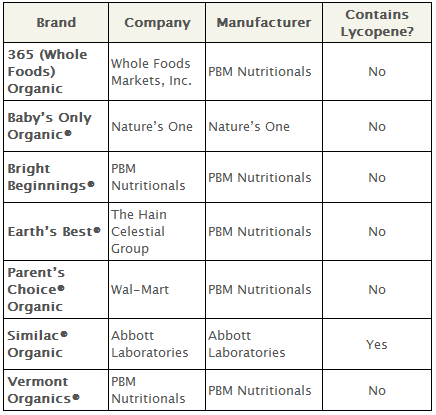
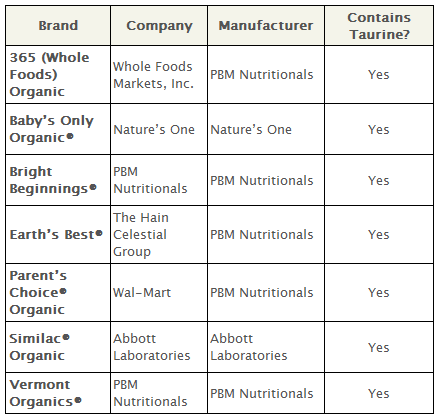
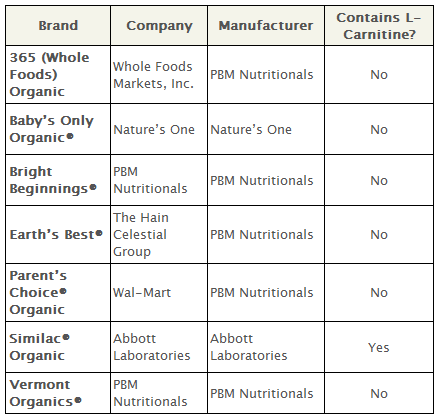
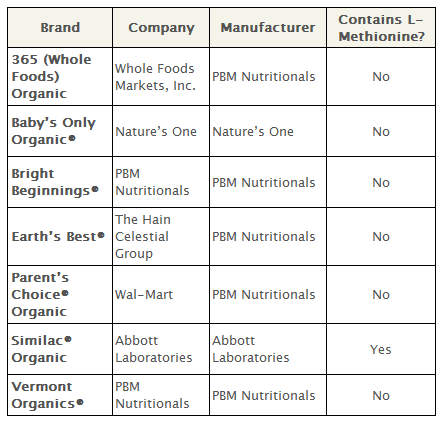
The National Organic Standards Board rejected the use of the following synthetic nutrients in dairy-based formula: lutein, lycopene, nucleotides, taurine, l-carnitine and l-methionine.
None of these nutrients are required in infant formula by the Food and Drug Administration, and all are prohibited in organic formula in the European Union. Some, like lutein and lycopene, are even prohibited in conventional infant formula in the European Union.
Lutein is produced from conventionally grown marigolds—likely treated with insecticides—and processed with the neurotoxic solvent hexane.
Lycopene is most commonly found in tomatoes, but the version in organic infant formula is produced synthetically by the chemical manufacturer BASF. A three-stage process is used to produce synthetic lycopene, and involves the solvent dichloromethane and the solvent toluene. Toluene is a neurological toxin derived from benzene.
Nucleotides are produced from hydrolyzed yeast. The yeast undergoes multiple chemical changes in order to extract nucleotides, including heating to denature proteins, cell wall proteolysis, enzymatic hydrolysis, and dehydration. The infant formula industry shared the identity of two suppliers of nucleotides for use in infant formula: one is a Chinese biotech company (Dalian Zhen-Ao Bio-Tech) and the other supplier is Japanese.
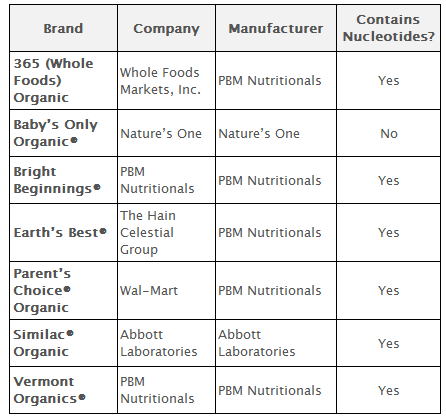
Taurine used in infant formula is produced synthetically; one processing method includes the use of sulfuric acid, a toxic and carcinogenic material, and another technique involves aziridine, listed as a hazardous air pollutant by the Environmental Protection Agency.
The production of synthetic l-Carnitine involves epichlorhydrin, a list 2B material (possible human carcinogen) by the International Agency for Research on Cancer. For this reason, it was rejected for use in organic foods by the National Organic Standards Board.
L-Methionine is required in soy-based infant formula to meet basic amino acid requirements. Given its incompatibility with organic principles, synthetic l-methionine is prohibited in European organic foods. For that reason, organic soy-based infant formula does not exist in Europe—another reason to avoid soy-based formula.
Soy-based formula is so nutritionally dissimilar from human milk that in some countries, like New Zealand, it is only available by prescription. Even the American Academy of Pediatrics has stated that soy-based formula provides an alternative to dairy-based formula only in very rare cases.
The synthetic version of l-methionine used in infant formula is produced with materials including acrolein, an EPA Hazardous Air Pollutant, and hydrogen cyanide, described by the Centers for Disease Control and Prevention as a “systemic chemical asphyxiant” and “chemical warfare agent,” “used commercially for fumigation, electroplating, mining, chemical synthesis, and the production of synthetic fibers, plastics, dyes, and pesticides.”
What’s A Parent To Do?
Parents naturally assume that the organic label means all ingredients were carefully reviewed, deemed safe and compatible with organic principles (these are requirements in the organic law). It is utterly disturbing that the infant formula and baby food aisle would contain some of the most egregious violations of the organic standards.
Even after the National Organic Standards Board voted to prohibit hexane-extracted DHA and ARA, carrageenan, two synthetic preservatives and six synthetic nutrients in organic infant formula, these unapproved ingredients remain in organic products on store shelves (lobbying efforts by the formula industry are apparently paying off).
Some parents make their own infant formula (Weston A. Price foundation has a recipe). Human milk sharing is becoming more popular as well, with social media making it increasingly easy to connect donors and recipients (check out “Human Milk 4 Human Babies” and “Eats on Feets”).
Other parents import organic formula from Europe, such as the Holle brand from Germany, since it does not contain any of the unapproved synthetic preservatives and nutrients (Holle still does contain palm oil and maltodextrin). In Europe, as in the U.S., these ingredients are prohibited, and manufacturers there follow the law.
Don’t babies in the U.S. deserve the same? When will the USDA’s officials in charge of overseeing the organic label stop bowing to the lobbying pressure of the infant formula lobbyists, and enforce the organic law?
Please Take Action and Share This Post
Let the FDA know how you feel about their decision to continue allowing carrageenan in our food, including organic infant formula. Sign Cornucopia’s petition here.
Writing this post was not easy. As a parent, I wish I could tell other parents who are in a bind and who need formula that the organic label signifies a safe option without any of the harmful ingredients found in conventional formula. As this post has shown, that’s not always the case.
That being said, let me stress again that organic infant formula remains a safer and a far superior alternative to conventional formula. Organic formula’s milk does not come from cows that were fed GMO feed, given antibiotics or injected with synthetic growth hormones. Organic formula’s sweeteners and oils cannot be GMO, treated with pesticides or extracted with neurotoxic solvents. So the choice between organic or conventional formula is a no-brainer.
Until we get unapproved ingredients out of organic infant formula, I hope this information will help parents make informed decisions when purchasing formula for their babies.
To view a PDF of the formula comparison chart in its entirety, click here.