Join The Cornucopia Institute as we keep you informed via web updates and live tweets from the National Organic Standards Board (NOSB) meeting online.
We will be sharing the play by play of public comments on April 21 and 23 and the meeting itself on April 29 and 30 below and with our Twitter followers at #NOSB or by simply following our stream.
For background on issues up for discussion at the meeting, see:
-
- Cornucopia’s formal written comments.
- Transcript of Kestrel Burcham’s oral testimony before the NOSB on April 23.
- Transcript of Anne Ross’ oral testimony before the NOSB on April 23.
Read Cornucopia’s notes on the NOSB meeting beginning April 29 at 11 am ET.
Find our notes from the April 21 public comments and April 23 public comments below.
You can listen to the meeting on Zoom–the link will be provided here.
Thursday, April 30, 2020
11 AM ET: Opening Roll Call
Ela leads the official roll call for NOSB members. All Board members are present at the start of the meeting.
[A list of the current NOSB members can be found here. NOSB members serve for 5 years.]
11:05 AM ET: Handling Subcommittee
Asa Bradman introduced the Handling Subcommittee agenda, starting with Handling material reviews.
Scott Rice begins with the L-Malic Acid (reclassification of L-Malic Acid) discussion document.


Rice: [Discussion concerning] moving from 605(a) to 605(b). Commenters noted that while supporting documentation may state that it is produced naturally it refers only to second half of the process. It’s the first part that gets sticky. To get acid 2 options for obtaining in first step: (1) fermentation of carbohydrate (non-synthetic); (2) as synthetic through malic acid of petroleum origin. Comments in response to questions: reclassification could affect listing of other substrates due to fermentation. It’s clear decision tree needs to be clarified. Commenters suggest Board clarify use of decision tree as applied to microbial products. [Predominant source is synthetic… It is not clear how much time is needed for other sources become available. Also noted some opposed moving L-Malic acid believing it first should be removed then re-petitioned or listed at 605(a) only for non-synthetic methods. Moving forward we need to come to decision on how to approach classification of fermentation byproducts. In order to get clarity, we need to revisit 633 decision tree as it relates to products like this and get a little more specific on starting point. More work to do in subcommittee.
Ela: Could you repeat, if we don’t go down the path of really revisiting classification tree then restate choices.
Rice: The most straightforward would be to move it to 605(b) and include annotation that the non–synthetic source be used when commercially available. That’s about as cut and dry as we could get.
Jeffery: We can’t afford to avoid fermentation to protect organic systems from new wave of GMO technology.
Ela: This came up with biodegradable mulch and where GMO technology leaves us.
Rice: Not opposed at all to diving into this just know that this is a larger conversation and will require some more input. We have useful comments on a path forward.
Bradman: My stance is that fermentation products is challenging but of concern and we should be thinking about that. For simplification, Scott’s cut and dry suggestion might help us deal with this substance, but need to think about larger issues.
Rice: Agree would be kicking can down the road. We have to deal with this at some point.
Baird: Echoing same concerns Asa just expressed. Any time you see anything produced with fermentation we are going to be raising issues with GMOs. Are we going to tackle it or ignore it.
Mortensen: Question is where is the boundary before which we are concerned about something in a process. At what point do we say this is allowable up to this point. Where do you draw the line on some of this. I think it would be helpful to have this discussion.
Jeffery: I was in retail location when GMOs received most attention. Now that I’m in another retail environment I see how far away organic is from helping customers understand what organic does as opposed to just being non-GMO. Technologies developing so quickly, the customer doesn’t und3rstand how much in the food system. If we can’t communicate to consumers, we have a big problem.
Seitz: I want to echo what Mindee just said. Two most common questions from customers: How do I know if GMOs and how do I know if grown with highly toxic pesticides?
11:25 AM ET: Handling Subcommittee (cont.)
The Handling Subcommittee moves onto the Discussion Document on Ion Exchange Filtration.

Ela: This issue came down from the NOP to try to discover a discrepancy between how certifiers and the program are looking at the issue. We have a column with attached ions and when corn syrup floats by they exchange ions; chemically filtering the substances at ionic level. Once those resins become clogged with impurities, do we just need to have recharged materials or resins themselves on the National List?
We list everything in the process? Or some things only? Are the resins considered a food contact substance? If so, they do not need to be on the National List. If they are secondary direct food additives, they do need to be listed. The FDA has listed them as both, causing confusion. OMRI found in 2002 that ion exchange is a chemical process, they are processing aids, and resins that have gone through this process of ion exchange are in fact a different product.
This process is used in the production of whey protein, infant formula, corn syrup, and other common products. The NOP seeks to resolve this complex conflict.
Bradman: Food contact materials can contaminate or affect food. Even if simply considered a food contact material, we should look at this. There is chemistry involved here, so it’s more than a food contact material.
Ela: It is a chemical exchange. BP noted this is similar to a water softener. I think the ions are relatively stable and literally the resins are essentially permanent; the ionic charge is what changes. There’s a good argument that resins aren’t part of chemistry except that they are the source of ions.
Greenwood: If we’re worried about contact of one chemical, then this needs to be looked at. For instance, phthalates are found in any water that has touched plastic. We need to be concerned about this.
Ela: Appreciate the program not making unilateral decisions and bringing this back to NOSB.
11:37 AM ET: Handling Subcommittee (cont.)
The Handling Subcommittee turns to the Fish Oil annotation change discussion document.


Bradman: In May last year, the subcommittee and then Board approved to relist fish oil on the National List but among Board members there was concern about exploitation of marine resources and implications on the environment. Some members wanted it to be consistent on addressing sustainability and over-exploitation. This was added to our work agenda. No one voted against relisting fish oil. Two members abstained.
I tend to think of this as a consumer choice issue. We aren’t really dealing with whether it should be allowed, but if we want to introduce environmental sustainability issues into the way the material is sourced. We have used 3rd party classifications to determine if the fish oil is sustainably harvested. Idea was to source from populations where over fishing is not occurring. There are also private marine stewardship standards in many cases nonprofits. From our thinking, almost all fish oil that comes into US is from outside US waters.
We received a number of comments on issues mostly from organizations like nonprofits and other organizations – very few comments from consumers. Some of comments from larger organizations say there needs to be clarification on whether from wild or farm raised. Some industry sources are concerned about loss of product and they don’t feel like NOAA or FOA definitions are appropriate partly because functionally they have different standards.
They may have different temporal assessments of populations. There are concerns about how ceritifier would deal with this. One certifier said they would ask for an affidavit about sourcing. The proposal as it stands could cause confusion in implementation. Some major producers harmonized their recommendation and none are against addressing issues of sustainability.
Oakley(?): There was good Support for “byproduct only” in the petition.
Bradman: Some of the omega 3s people want to add to their diet are also available from algal oils. Potential non-fish source of these fatty acids.
Baird: We’re seeing this in crops, livestock, and now in handling: the same issues of sustainability of marine materials. Perhaps one way would be to address them by a new definition, including what we consider to be sustainable harvest of these crops.
Mortensen: There are dependent aims here. Yesterday we were discussing the merits or lack thereof in paying attention to the harvest of fish when the alternative is oil. The argument feels almost circular. We need to determine whether fish harvest is occurring for fertilizer only.
Bradman: The point about plant sources is important. During oral comments I mentioned plant sources and their concern was that some of the precursors to Omega-3s do not exist in sufficient quantities to supply what is needed by the human body.
Oakley: We’re looking at meal in livestock feed as well. If we parse them individually for harvest, that may be simpler, but it doesn’t address the issue of the three products (meal, oil, and solubles) from fish.
11:55 AM ET: Handling Subcommittee (2022 Sunset Review)
The Handling Subcommittee now moves on to a review of the handling materials up for Sunset Review (2022).
Kaolin:
Huseman: Kaolin. There were very few comments. Kaolin is aluminum silicate. A couple certifiers said a couple handlers are using it. Would be interesting to know if there are alternatives. It is also produced in nano-particles and suggestion to prohibit nano kaolin.
Sodium bicarbonate:
Jeffery: Certifiers had a lot of comments. Substance being using ubiquitously. ACA would support reclassifying as synthetic. The substance is essential across many categories and general support for relisting it.
Waxes – nonsynthetic (Wood resin):
Huseman: This is used for fruit coatings, one of few options available for use. Comments include questions and concerns. Most commenters want to ensure use of this material. Some comments suggest volatile synthetic solvents may be in use for production. Should there be labeling noting there is wax on the product for consumers? Relisting is overall supported.
Bradman: Agrees there should possibly be some sort of labeling here. Wax cover on fruit cannot be removed and constitutes an unlabeled ingredient.
Ela: Notes his apples are not coated. This covering keeps them shiny for longer than just polishing the naturally occurring pectin.
Oakley: It is an unlabeled ingredient. Interested in the further discussion.
Baird: Points out we cannot change annotation during sunset review. We would have to do it later. Opines it has to be a voluntary labeling. If we required labeling of waxed product, I think it would be challenging. We would have to go into the labeling section of the regulations.
Ammonium bicarbonate:
Jeffery: Not a lot of use for some of the certifiers. Others said it is commonly used. There is support for relisting. Important as leavening agent. Beyond Pesticides said candidate for delisting because of emissions of ammonia and carbon dioxide.
Ammonium carbonate
Jeffery: Less info for this. Same concern about emissions. One support for relisting. Similar comments as for bicarbonate, but fewer comments.
Ela: Are there distinct uses?
Jeffery: I’m just seeing as a leavening agent.
Ela: It’s always good to get comments.
Calcium phosphates (monobasic, dibasic, and tribasic):
D’Amore: Total of 16 public comments, only one strongly opposing. Concerns about cumulative effect phosphates for human health. Concerns about unreliable, consistent alternative and availability of organic version.
Ozone:
Rice: We did get info that 51 operations use it. It is generally regarded as safe. One comment expressed concern about its use in irrigation water. Overall, it is highly regarded and commonly used.
Greenwood: I have ozone for the 2023 sunset for water sterilization. I found a 2002 TR. Do you think we have enough information or should I request a TR for 2023 review.
Rice: We did suggest NOP contract an updated TR before the next review.
Greenwood: I’ll request it to see if any new information.
Bradman: It is an air pollutant in general populations. This like other materials for food safety there needs to be adherence to standard procedures to protect workers. Just a reminder that some of these materials have potentially serious occupational health concerns.
Ela: Stakeholders ask us to look at food safety and environmental issues. We were going to have a panel discussion and planning to have one in the fall if we have a live meeting.
Sodium hydroxide:
Jeffery: General support for relisting. Beyond Pesticides noted annotation supports investigating essentiality.
Carnauba wax:
Huseman: Another wax that can be used on fruits and vegetables and on gummy bears and jelly beans. Several comments saying that commercial availability is sufficient. Over a dozen suppliers. Some countries only allow certain formulations of waxes so having the ability to use one of three allows for export to different countries. It might night be as effective in some applications as in others. A certifier said several handlers that list this product that list in the organic system plan. Overall some mixed reviews.
Ela: When looking at processor using it are you looking if its organic or synthetic (to Scott and Nate)?
Rice: Yes. We look to see if there is an effort to source organically.
Powell–Palm: I concur with Scott. Sourcing will be affirmed upon inspection.
Turner: When thinking about sources, for example when a material comes from Brazil or other places. We need to think about what involves getting that material to market.
Huseman: When speak to sustainability that should be addressed at least.
Glycerin (CAS # 56-81-5):
D’Amore: 18 public comments, largely supporting renewal. The key question was about the 99% pure designation. Is the current supply of organic glycerin truly immature, or is it being undermined by relisting this non-organic version on 606?
Bradman: There is a lot of concern about 606 preventing the production of organic materials. Wish we had criteria to evaluate this concern.
Inulin-oligofructose enriched (CAS # 9005-80-5):
D’Amore: 23 public comments. [Comments were mixed.]
Some said alternatives would be widely available. As I joined the team, I was overwhelmed by the science and now I know its knowable if spend time looking at it. Noted personal sense of wanting to re-list tools but will seek information about the materials under his purview.
Baird: I think we’re all conflicted on the Board and all come in a little intimidated by the science. We all want to protect tools for farmers and maintain organic integrity. We’re all there with you.
Rice: It is a challenge with materials, but on certification side we are going to looking for organic version or material. We have seen success stories with 606.
Colors (18):

Ela: Colors have a lot of nuance. There are 18 materials included here. Previously, many were suggested to be de-listed by subcommittee, but the full board discussion brought up questions of availability. Some of the crops have to be specifically grown for colors; it is not just a crop byproduct. The incentive is based on price. Comments conflicted here. IFAC committed to giving more detail to the Board. Plans to contact manufacturers before fall.
Beet juice extract has conflicting data. One company says we have sufficient supply, others say not. Conflicting data from multiple manufacturers on availability of many colors. Will continue to explore the supply of each and possibly de-list some of them. There may not be enough organic production to de-list others.
Mortensen: Would vote on individual colors in the fall?
Ela: Yes, that is the plan at this point.
Mortensen: If suppliers and users are finding adequate sources, that would be an important consideration for de-listing. Re: beets – 95% of conventional beets are genetically modified. Mindee’s point earlier about source material considerations should be considered in this category.
Ela: Some comments note that organic is far more expensive than conventional, but that is not how relisting is determined.
Bradman: Working on a risk assessment on FDA artificial food dyes. Not a conflict; not profiting. Colors are not essential, although they have a lot of purposes. There was argument that some organic sources didn’t yield same quality colors as conventional or achieving same hue. Why can’t we have different hues in organic? Can they contract for some of these colors? In terms of cost to the food, it seems unlikely it will drive cost up substantially. Doesn’t quite understand the obstacles to producing thee from an organic source
Ela: Literally all organic grapes would have to go into colors to produce a supply of color. Some of these are truly needed for consumer acceptance, and others present the issue of ease of use for manufacturers only.
D’Amore: Can we put varying dates on the sunset period, as they show promise for an alternative?
Ela: The sunset period is five years.
Rice: When we look at time frames, such as we attempted with methionine, it creates significant challenges to the program.
Tucker: If the Board voted to remove during sunset, the NOP could put an implementation timeline for implementation.
Bradman: Would like to hear more from stakeholders about use of colors as liquid versus powders. Is this a factor in whether we’re using organic or conventional material?
Ela: This came up last time around as well. Hoped to get more information in comments. Inclined to propose de-listing unless we get other input.
Kelp:

Romero Briones: A lot of public comments. She has more than 75 comments. There were references in fish oil and proposals for marine materials. Generally, hard to say if majority if for relisting or de-listing. Comments on both sides. Comments from NOC were really helpful. Referenced listing of kelp under 606 and that NOSB has a responsibility to look at impacts on ecosystems and numerous seaweed and kelp listed separately. A lot of comments said “kelp” is too generic. IOIA had wonderful diagram and did give a great overview of what is considered marine algae and kelp. We’re specifically reviewing 606 listing. Comments mainly focused on ill–defined term of kelp. NOC suggested de-listing under 606 so there is a greater chance of monitoring ecological impacts. General sense that kelp is not well defined.
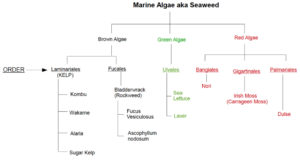
Oakley: What is your sense of it there are sufficient organic supplies of kelp for human consumption?
Romero Briones: There was a comment that referred to regeneration of kelp and that it occurs pretty fast. Other commenters said need to de-list because hard to monitor consumption and gathering methods because it’s listed under 606. NOC commented that if we de-list from 606 will have a better idea whether gathering techniques are ecologically sound.
Oakley: Last fall when we were relisting some seaweeds, it was complicated. We should do more research before the vote in the fall.
Romero Briones: It is so complicated and not a clear understanding how we’re handling kelp and how we give permissions for specific types of kelp listed separately.
1:05 PM ET: Lunch Break
The NOSB will return at 2pm ET.
2:00 PM ET: Handling Subcommittee (2022 Sunset Review, Cont.)
Starches: Cornstarch (native):
Bradman: Cornstarch may be available in organic form; still to be determined. Commonly used. Stonyfield notes some organic cornstarch did not work in yogurt production. There are different forms of this product (8+ versions). Seeking clarity regarding why it didn’t work and what is needed to make it work.
Regarding dressing and sauces, no market analysis has been offered to justify notes that there isn’t enough organic available. Comments noting that supply consistency may be unreliable. Some forms of cornstarch are essential. Some trade organizations have looked at the number of acres of certified organic corn in the US and found the acreage too low to provide source material needed. The production is also commonly too far from manufacturing facilities to be workable.
There are suppliers that note they produce a lot of cornstarch. Others think cornstarch should be removed because there are other organic starches that can take its place. May include annotation for specific kinds of cornstarch (i.e. high amylose).
Mortensen: This is an example of a non-perishable product that seems to be track-able to make this work off the 606 listing.
Bradman: All producers note they are non-GMO certified to help ensure these are not genetically modified. One comment noted costs are higher for organic and yields are typically lower, creating a bottleneck for availability. Cost is not NOSB’s main issue here.
Powell-Palm: Experienced inspector and grain producer. Doesn’t perceive this as a bottleneck issue. US is good at growing organic corn with high yields. This is a great opportunity for farmers.
Baird: The reason we’re having so many problems with fraud is because we are limited in our organic corn supplies. To sort the specific types at a processing facility is a hardship. Processors are generally unwilling to stop normal production to make a small run of organic product.
Starches: Sweet potato starch:
Bradman: Similar to cornstarch. There is some support for removing from the National List, but more market information and location of production (perhaps East Asia) is needed.
Orange shellac-unbleached (CAS # 9000-59-3):
Huseman: Falls under the same type of category as type of product used to poach fruits. Produced through secretions of an insect. It is used in the jelly bean manufacturing process. Commenters stated alternatives are not sufficient because have not proven a functional equivalence. Most comments were for relisting. The 2014 TR mentioned a number of ancillary substances and questioning which ancillary substances are allowed to be used on organic produce. For the most part comments support relisting.
Turkish bay leaves:
Rice: Turkish bay leaves were unanimously voted to be removed at the last sunset review. A manufacturer noted that the product they needed was not available and it was relisted. Numerous comments support the removal from 606. Barring any further input, it seems we will move to relist in the fall. This is potentially a 606 success story.
Mortensen: If Turkish bay leaves were not on 606, and a producer was unable to find the organic product, could they use conventional with certifier concurrence?
Rice: No.
Turner: Has the producer who intervened last time weighed in this time?
Rice: I cannot answer that because it was listed in Organic Trade Association (OTA) comments.
Turner: Just wanted to raise the question, as doesn’t understand how that happens.
Rice: Generally, the sunset process works, using two comment periods. In rare instances like this one, we see an operation step forward after the comment period, or where comments were not persuasive. The rulemaking continues after the NOSB, through the USDA. Even one comment can be persuasive to the USDA.
Turner: Thinks this is a good reminder of the process for himself and stakeholders.
Ela: A few products over the past few years have been derailed from de-listing due to a single comment. Comments of NOSB do not carry through the entire rulemaking process. It’s important to stress the number of comments NOSB receives and give further information to the NOP/USDA other than just stating the decision made.
Whey protein concentrate:
Romero Briones: Received approximately 30 comments although more when consider form letters. Points out Harriet Behar’s comments who went through a very detailed history, specifically about the votes in previous NOSB reviews about delisting. She said she doesn’t’ understand why it hasn’t been delisted. The large majority of comments were for delisting. Another notable commenter was CROPP and they are a major supplier of whey protein concentrate. CROPP said they produce so much it spills into the conventional market. Several certifiers said 6 or 7 producers included this in organic system plans. We do have a petition to delist which is whole separate process.
Ela: Found it interesting CROPP said had a large supply, selling some on conventional market. I found that to be a strong comment.
Mortensen: I agree with Steve. It also strikes me that when NOSB voted unanimously to remove. Economic restraints on dairy farmers would support delisting. I would hope we could do what we could to support organic dairy industry.
Romero Briones: 606 can be seen to a detriment to organic production or a safe house until that market is more robust. I think whey protein concentrate has outgrown 606.
Oakley: I was wondering what prospects of delisting might be?
Romero Briones: In Harriet’s comments she does note that she doesn’t understand breakdown in the process. NOSB has suggested in the past that it be delisted. This is an area that is not quite understood.
Bradman: Maybe the program [the NOP] could comment on it.
Jenny Tucker: Always remember this is a 2–phase process. NOSB sends recommendation then NOP does rulemaking under Administrative Procedures ACT and Economic Impact Assessment. So the two–phase process means different things are considered in each step and the public has the opportunity to comment along the way. Both parts of the process are vitally important and need to balance one another. Could be that rulemaking results in something different than NOSB recommendation.
This concludes the Handling Subcommittee material review.
2:38 PM ET: Livestock Subcommittee (Fenbendazole)
Sue Baird starts the Livestock Subcommittee discussion with a petition that was turned into a discussion document.
Discussion document: Fenbendazole – petitioned 2022 (for use in poultry):

[Cornucopia does not support the listing of Fenbendazole for use in poultry unless the term “emergency” is strictly defined. Right now there is no defined “emergency use” which will lead to fraud and confusion for certifiers. A withholding time should also be required for both eggs and slaughter stock. Management strategies are the best way to control dangerous worm loads. There are rare situations where this tool has legitimate use to protect animal welfare.]
Baird: Fenbendazole currently listed for emergency treatment for dairy breeder stock during specific stages of life only. Annotation amendment has been requested for laying hens. [Describes history of discussion of use in treating broiler chickens and replacement chickens, laying hens, and breeder hens.] FDA has established the amount safe for use over animal’s lifetime. They determined that 2.4 ppm residue of Fenbendazole in eggs is safe. Studies suggest that residue is well below the safe level in eggs with no withdrawal period. This is how it was approved in conventional production. It is typically administered to conventional layers before they receive outdoor access and again after they contact the soil.
Fenbendazole withdrawal period was reduced from 90 days to two days for cows in 2016. This is for emergency use only. Large organic flocks with outdoor access are being infected with parasites at a higher rate than conventional, resulting in 9-18% mortality. High doses of liquid oregano improve prevention, but do not take care of an infestation. Worms in eggs would affect consumer belief in organic label.
[Note that the 9-18% mortality due to parasites in organic flocks referenced is likely because the majority of organic egg production is industrialized without parasiticides as a tool. However, these industrialized egg producers that make up a majority of the egg industry and are “faux organic”. Authentic egg producers rotate their birds frequently, which significantly reduces parasite loads without the use of any chemical inputs. For more information on the organic egg industry check out Cornucopia’s report: Scrambled Eggs: Separating Factory Farm Egg Production from Authentic Organic Agriculture.]
Four kinds of comments received:
Human health concerns regarding residue in eggs at zero withdrawal time. Would we start seeing parasite resistance to Fenbendazole in humans as we have with antibiotics previously routinely used in poultry? Fenbendazole is used for human treatment of human parasites—would this result in human parasite resistance? Comments note that organic production has never been guided by conventional. Fenbendazole residues have been detected in eggs through the eighth day of treatment.
This suggests 14 days of withholding: 5 days for treatment, 9 days for withdrawal. Another issue is that these birds can end up as slaughter hens, and all parasiticides are prohibited in slaughter stock.
Producers report a large uptick in worm infestations.
Seitz: Reluctant to vote on this because once something is voted on, it requires two-thirds vote to remove. Concerned about non-targeted use probability. A number of certifiers said this is not needed. Cultural methods are available to prevent infestation. Concerned about residue in eggs—uses raw eggs in mayonnaise. Felt a lot of sensationalism was employed to persuade the Board—has never seen worms in eggs.
Powell-Palm: Do you have information about correlation between the square feet of outdoor access allowed and the parasite load?
Baird: Did not see that.
D’Amore: Is it possible to have an organic egg?
Baird: Yes, but petitioners say this has become a much bigger problem as we give them more outdoor access.
D’Amore: The astounding thing to me is the tolerances built up to Fenbendazole. That was the biggest thing I heard.
Mortensen: Bothered by the notion that this is organic IPM for chickens. The argument was that this needs to be a routine kind of cleansing of the birds, raising questions about the foundational cultural management system. Appreciated NOC’s comments on this. We cannot have an emergency every time there is a new flock in the hen house. I heard this same argument 20 years ago in conventional agriculture for herbicides. If there is no feedback to cultural practices, this is problematic.
Baird: NOP has not accepted any definition of emergency. Canada has said it can be used once in an animal’s life. Waiting for the TR; may be just guessing until then.
Bradman: “Emergency use” can be abused. Must define that term. You’ve implied a withdrawal period isn’t workable. Japan does have a withdrawal period. Is it feasible?
Baird: Producers in Missouri say that they contract for their eggs and contracted eggs cannot be diverted to conventional markets. They also have to administer to the whole flock or none. One producer reported getting 19 cents per dozen. It does not seem to be a workable solution to employ withdrawal times. [Then notes that the “size” of these typical producers are bird houses of 8000 birds in it and 3-4 houses—which is why they think this is unworkable if they have to withdrawal times for such large flocks.]
[Cornucopia notes: If the house has 8000 birds in it and 3-4 houses as referred to here in the discussion—that is not authentic organic production. Authentic organic egg production is found on rotated pasture, not in massive crowded flocks with less usable space provided outdoor than in. The declared need for Fenbendazole in poultry is clearly tied to industrial production rather than a true understanding of what might constitute an “emergency” for an authentic pastured poultry operation. This makes it clear that without withholding times and “emergency” clearly defined, fraud and overuse would be inevitable.]
Romero Briones: Hard due to lack of understanding of livestock practices or effects of fenbendazole. I would vote not to list, using the precautionary principle.
3:20 PM ET: Livestock Subcommittee (2022 Sunset Review)
Butorphanol:
Rice: Treatment for pain prior to surgery for animals. Majority of commenters supported relisting. Important but not a lot of operations using it. Several dairy organizations advocated for its continued listing for welfare of animals. One organization noted that more info about metabolites when excreted needs to be evaluated. Alternatives are out there but not as effective. Similarly, herbal remedies are available but not to prevent acute pain.
Flunixin:
Baird: It is a compound that is potent non-narcotic and analgesic agent with anti-inflammatory activity. In public comments, 19 organizations commented and all of them said “yes.” Beyond Pesticides said just use aspirin and did note that residual effects are prohibited for horses for slaughter.
Poloxalene:
Seitz: Used to treat bloat. 10 comments in favor of relisting. A couple comments questioned whether there is a natural or non-synthetic approach to treat bloat. Even if needed rarely, it is essential to producing.
Formic acid:
Powell–Palm: Used as external pesticide within honey bee hives as well as local anesthetic. Overall those who commented were in unison that it should be relisted. In depth comment from Harriet that organic practices by bee keepers should be encouraged until there is a standard.
EPA List 4 – Inerts of Minimal Concern:
Rice: Had substantive discussion yesterday with crops subcommittee. Don’t know that we need to dive in this too much here since looking at the same thing. Reiterate that there is universal dissatisfaction. Repeated recommendations from the Board over the years with a roadmap as well as comments NOP did not take it on. NOP seems ready to take it on so can finally move forward. Reiterate Dr. Tucker’s cautionary note that if we delist, we need to keep in mind OFPA does not allow addition of materials outside of Board’s actions. To avoid disruption in the market place may need to add material.
Excipients:
Baird: Allowed only for use in manufacturer of drugs only if FDA recognizes it as safe and approved by FDA approved as food additive or part of new drug application. Used in new animal drug applications and animal health care products. Common in almost all therapeutic products veterinarians use. Can be natural or synthetic. Derived from GMOs or by other means. They can be simple or complex. Got a lot of public comments. Certifiers are interpreting differently and allowing different leeway. NOC did incredible job summarizing the interpretations and inconsistencies and how to regulate. Issues concerning certifier differences in allowing direct and/or indirect food additives. Most certifiers say they are using best practices document that ACA developed because of confusion. PCO: discrepancies do a disservice to our operations and clarification is needed. This is problem we need to address. Some certifies say address individual problem children (excipients).
Strychnine:
Powell-Palm: Consistent support for relisting. One point brought up is the ripple effect of organics. Strychnine moves up the food chain, and anything that consumes a rodent killed with strychnine will ingest it.
Magnesium hydroxide:
Buie: Been on the list for over 15 years. Comments overwhelming in favor of keeping it. Past review, Board did not recommend removing from the list.
This concludes the Livestock Subcommittee portion.
3:37 PM ET: NOSB Work Agenda
[Note that the sanitation materials panel will convene in fall. It was postponed from this meeting in order to occur in person.]
Fall NOSB meeting to occur October 27-29, 2020 in Cedar Rapids, Iowa.
Spring NOSB meeting to occur April 28-30, 2021 in Crystal City, VA (tentative)
Tucker: Thanks to Steve Ela for his leadership. On a personal note, lives two blocks from the meeting place in Crystal City, VA. Acknowledges all board and audience for community collaborative engagement.
Ela: Shares “For The Children,” a poem by Gary Snyder:
The rising hills, the slopes,
of statistics
lie before us,
the steep climb
of everything, going up,
up, as we all
go down.
In the next century
or the one beyond that,
they say,
are valleys, pastures,
we can meet there in peace
if we make it.
To climb these coming crests
one word to you, to
you and your children:
stay together
learn the flowers
go light
Wednesday, April 29, 2020
11 AM ET: Opening Remarks
Dr. Jennifer Tucker, Deputy Administrator of the National Organic Program (NOP), welcomes all to the Spring 2020 NOSB meeting. This meeting is being held remotely due to the impacts and closures from the COVID-19 virus.
Bruce Summers, Administrator for the USDA’s Agricultural Marketing Ser
vice (AMS) makes a statement at the start of the meeting:
Employees that can are working remotely; but it’s important to note that vast majority of AMS employees don’t work in an office [anyway]. They work in food processing facilities, ports, farms, grain elevators, etc. Thousands of AMS employees they are out on the lines and collecting critical information. AMS Market News is a unique thing. That data that is given voluntarily by stakeholders is critical in getting the Secretary, Congress, or White House the info they need [in this time]. AMS is operating, but it’s not business as usual. Still, AMS is working really hard to maintain level of service (while still careful to follow CDC guidelines).
Summers notes pride in NOP for work on Strengthening of Organic Enforcement Rule and Origin of Livestock.
AMS purchases food – one of its signature programs. AMS normally buys this food for school lunch; in recent years has gone up a bit like surplus commodities to a little over $1 billion in 2018. Almost a billion a year is spent moving food into supply. With the closure of food service industry around the country, and disruption of supply chain there is a lot of food having trouble getting into retail sector.
AMS will start purchasing $300 million of food each month and work with food service sector to use logistics power to procure food, pack it and deliver it to nonprofit organizations. AMS has never done anything like this before. We hope to have contracting process worked out this week and shipping by middle of May.
AMS is working across department to work on CARES act legislation which calls for direct payments to producers. Hope to have it published soon and implementation to start by end of May, including small local and regional and organic. Hopefully no farmers will be left out of that program.
Things are more complicated for specialty crops because typically not involved in these types of programs. AMS values the NOSB’s recommendations.
Tucker then introduces Steve Ela, the chair of the NOSB and the NOSB’s meeting facilitator.
11:18 AM ET: Questions from the NOSB for AMS
Ela turns to the NOSB members to see if they have questions for Bruce Summers.
Emily Oakley: Are there steps for purchases from producers having trouble accessing federal money?
Summers: The BOX program [The Farmers to Families Food Box Program] – we are working to set up in a way that small, local, and regional can supply food into that program. Expecting that food hubs will submit proposals to work on distribution side.
Nate Powell-Palm: Thank you for mentioning Origin of Livestock. The entire organic industry is eager for final rule and to clarify and clean-up the livestock component of dairy sector. Hoping the rule goes to final rule and able to experience a clean–up of over-supply of milk.
Summers: The Origin of Livestock will be final. Jenny will give more details on exact status of that rule. It’s on track.
Scott Rice: On the Strengthening Organic Enforcement rule – I understand it’s in final clearance? Want to reiterate importance of the rule. Certifiers are looking at different ways of conducting work. Jenny’s team doing same with accreditation oversight. Hopefully [this rule will] come out despite challenges with pandemic
Summers: Doesn’t think COVID will slow [Strengthening Organic Enforcement rule] down. It’s going to be a bit of a race to see which one comes out first. The Secretary supported this rule. These rules are moving forward.
Ela: Farmer’s Markets and specialty crops producers – anything the department can do to make sure these markets are [considered] essential? Having a market closed drives people back to grocery store; need to keep markets safe and open or [there will be a return to] to grocery store model. Anything you can do is greatly appreciated.
Summers: Heard innovative stories about markets staying open – like additional hand washing facilities. We have USDA farmers market and AMS is responsible for managing it. It’s closed right now because most people in DC working remotely and tourists are there. As we look to what’s going to happen this summer, we’re looking what we can do with USDA farmer’s market that would allow people to keep shopping. We view USDA farmer’s market as a laboratory. We hope to provide some leadership.
Ela: If you could highlight what is and what isn’t working that would be helpful.
Summers: AMS is looking at different grants, cooperative agreements, and what we do with universities so we can help fund research on best practices. Looking at rapid response studies that can make available to local and regional and primarily organic sector. AMS looks at agriculture across the whole board and looking at ways to work with farmers markets and food hubs and way to provide resources immediately, not a year from now.
[Cornucopia’s take on Origin of Livestock: Despite overwhelming support in 2015 for closing this loophole via the proposed Origin of Livestock Rule, the USDA has delayed in publishing a final rule. As it languishes, authentic organic dairy farmers are being undercut in the marketplace by factory farms whose certifiers interpret unclear regulations to their economic advantage. As a result, many authentic organic dairies have closed their doors. Most organic consumers are unaware of the bait and switch. Cornucopia hopes the final rule will close all loopholes and save authentic organic daries.]
11:30 AM ET: Introduction of the NOSB Members
Ela leads the official roll call for NOSB members.
[A list of the current NOSB members can be found here. NOSB members serve for 5 years.]
Sue Baird: From Missouri. Executive of mid-america food hub. Many years experience in organic world. 4th year as NOSB member.
Asa Bradman: Professor at University of Berkeley. Works in issues affecting health and children. 4th year as NOSB member.
Jessie Buie: Producer in 5th years of service on NOSB.
Rick Greenwood: Professor at UCLA and CA organic avocado grower for about 20 years. Is on CA governor’s testing task force – trying to figure out ways to get things back to normal.
Jerry D’Amore: Brand new NOSB member. Specialized in berry crops. Prior to that operated hydroponic farm. 1st year NOSB member.
Kim Huseman: Director of Specialty Ingredients for Pilgrims. 1st year NOSB member.
Mindee Jeffery: Retailer; 1st year NOSB member.
Dave Mortensen: Serves in scientist seat. Faculty member and chair of agriculture and nutrition systems dept at university of New Hampshire. Work has focused on on-farm and farmer participatory research on organic and sustainable systems. 4th year NOSB member.
Emily Oakley: Farmer representative. Grows diverse organic vegetables and sells directly to consumers. In 5th and final year as NOSB member.
Nate Powell-Palm: Lives and works in Montana. Currently in thick of planting spring grain. Eager to turn cows out on pasture. Certified organic since 2008 and runs a first–generation grain and cattle operation. 1st year NOSB member.
Scott Rice: External coordinator for WA state dept of agriculture.
A-dae Romero Briones: Work for First Nations Development Institute – works with Indigenous producers across the country. Sits in consumer seat and in last year on Board (5th year).
Dan Seitz: Public member/consumer representative on Board. Lives in MA. Regular work is executive director of Counsel of Naturopathic Medicine and also on board of food coop. Been involved in natural health movement for years and understands importance of real food and building healthy immune systems. Background in law and higher education. In 5th and last year on NOSB.
Wood Turner: Senior VP . Trained as environmental and landscape planner. Grew up in NC and now based in CA.
Steve Ela: Farmer seat; fruit grower in Colorado. 4th year as NOSB member.
11:41 AM ET: NOSB Report
Jesse Buie, NOSB Secretary, gives the Secretary’s report: Fall 2019 NOSB Meeting minutes accepted as written.
Steve Ela, NOSB Chairperson, gives the NOSB report:
Five chairs are opening at the close of 2020: 2 farmer, 2 public interest, one accreditation seat.
[The process for nominating NOSB members is an important part of fair and accurate representation of organic stakeholders within the organic label.]
Encourages capable people to apply for seats; losing board members that have given great diversity to the board. Seeking diversity as nominations are considered to provide voice of undeserved communities.
Inspections and certifications that rely on in-person meetings are impacted by the coronavirus crisis. Farm insecurity is now felt throughout the industry. Organic agriculture is based on resiliency, but there remains a tremendous amount of insecurity in the marketplace. Asks the NOP and AMS to continue to support organic producers. We hear a lot of division in these meetings; the NOSB is here to argue out the gray areas. Our tax money is paying for these important conversations, and it would be very difficult to fund and carry out this work without the NOP. There is no black and white between organic and conventional. It is our job at the NOSB to draw the distinction in the gray area. We may not always hit the mark exactly, but I think we do a darn good job.
Comments on the rule-making process make a big difference. Encourages all to comment on proposed rules.
There is only one vote required in this meeting. This is unusual, but will be made up for in the fall meeting.
Ela then thanks the board and stakeholders for their presence.
11:55 AM ET: NOP Report
Jennifer Tucker gives NOP Update:
Organic Integrity Learning Center is moving forward with more training available.
Encourages people to pay attention to the USDA page for updates on coronavirus issues [this can be found at: https://www.usda.gov/coronavirus/ ].
The NOP is fully operational and teleworking, including desk audits. Requesting electronic submission of paper documents at this time. Encouraging critical thinking about how to ensure compliance during this period. The initial on–site inspection is a fundamental requirement and a virtual inspection cannot replace. Once certified, ongoing compliance may be ascertained via more creative approaches.
Strengthening Organic Enforcement: Proposed rule is with the Office of Management and Budget (OMB). Once they agree it’s ready to go, it will go to the Federal Register for comments. Input is needed!
Origin of Livestock: The draft of the final rule is nearing completion and is headed to the OMB next.

The electronic version of the NOP Import Certificate has been released and is currently optional. This allows the NOP to access data and build analytical tools. Pilot studies are in the near future. Once Customs and Border Patrol implements the import shipment filing system, other technologies must come online to implement.
NOSB call for nominations: The submission deadline is June 1, 2020 for the five vacancies.
[Note that the definition of the “farmer member” of the NOSB has been changed. Now instead of just a farmer who “owns or operates a farm” this definition includes “OR employees of such individuals.” This is problematic because farmer stakeholders cannot be properly represented by employees, for example, of a large “farm” corporation.]
We have continued to grow the NOP staff and program with additional resources. Have reoriented the program with two new divisions: International Activities and Trade Systems. These used to fall under Accreditation. Four employees were on-boarded virtually on Monday. Goal is to reach 59 staff members.

Oakley: Heard confusion in written and oral testimony that the three-year transition period is still causing confusion; shared some of the concerns regarding the requirement for farm sites to go through the three-year transition with no prohibited substances applied in this period.
Tucker: Agrees that certifiers use the handbook and memos to ensure this is the case. Believes those regulations are being implemented.
Seitz: This issue of container production has been very divisive and does not believe the divide will be bridged. Understands the NOP wanted to wait to work on guidance for this type of production, but this appears to be the time to determine whether hydroponic standards may be needed at this time. These standards may help to heal this divide.
Tucker: Agrees the topic is contentious and it has been discussed by the NOSB. Lawsuits are a tool by which policy is accepted or rejected. There is a lawsuit currently in play. The outcomes of lawsuits can provide clarity, but the courts will determine that. This topic is not on the agenda, but the NOP continues to take feedback.
[The lawsuit Jennifer Tucker is referring to deals with the USDA’s decision to allow hydroponic operations to be certified organic despite organic standards that require that farmers foster soil health (hydroponic and aquaponic systems do not utilize soil). Center for Food Safety (CFS), along with a coalition of organic farms and stakeholders, filed this lawsuit challenging the USDA’s decision in March, 2020.]
Powell-Palm: During public comments, the dairy compliance project came up. How are unannounced inspections linked to this project being carried out in the time of coronavirus?
Tucker: This is the third year of this project and we started early. Desk reviews of Organic System Plans are underway. NOP has staff located in areas of the country that will be able to travel as restrictions are lifted. There are inspections going ahead right now. Has confidence in the program moving forward this year to protect the market and ensure fairness.
Mortensen: You’ve indicated the board has discussed the issue of container production, but there has been no coordinated discussion addressing the criteria we’d like to see farmers follow in soil and liquid-based production. We’ve had a number of thoughtful comments from the public regarding substances used in production we would describe as highly soluble and there have been rules in the past restricting the use of these inputs. Not sufficient discussion or vetting of hydroponic methods since the allowance. This would be beneficial.
Tucker: These are important questions that continue to be raised in this setting. In the world, top priority of farmers is enforcement of existing rules, tackling issues such as fraud.
Mortensen: Thankful for work on the issue of fraud. Not comfortable with the notion that producers are not concerned with the issue of container production. The NOSB could take on the background work enthusiastically.
Tucker: We have a robust set of regulations that guide certifiers. Appreciates ongoing feedback from the Board.
Greenwood: Raised issue of more residue testing. Can we do more and randomized residue testing to ensure better enforcement?
Tucker: Testing is generally done to substantively support enforcement actions. We oversee the certifiers’ efforts at risk-based residue testing. Additional testing was required of Black Sea operations, for instance. Looking at how the NOP could do more sampling and testing of grains. Keeping a close eye on how certifiers are implementing residue testing.
Greenwood: Risk-based testing yields more positive results, but believes more random testing may prevent fraud.
Tucker: Unannounced inspections often include testing as well. Sampling and testing are important, and the Organic Integrity Learning Center will launch a course on how to ensure testing can stand up in court.
Turner: Acknowledges previous comments by Dave and Emily. Supportive of Dave’s suggestion and hopes the program will be receptive moving forward.
Tucker: Thank you.
[For more information on the issue of hydroponics in organics, check out Cornucopia’s report: Troubling Waters: How the USDA and Hydroponic Agribusiness Diluted Organics by Sanctioning Soil-less Growing. Cornucopia also has further information on the benefits of native ecosystems, including those that rely on soil health.]
12:38 PM ET: Compliance, Accreditation, & Certification Subcommittee update
Scott Rice, Chairperson, gives the Compliance, Accreditation, & Certification Subcommittee (CACS) update to the NOSB and listeners:
CACS does not have proposals or discussions to bring forward at this meeting. We have had a lot of discussions around COVID-19. Also wants to touch on certification community’s response to the pandemic. We saw markets disappear overnight. It has been amazing to see immediate response of certifiers and others to keep communities fed. Certification community saw that with social distancing in place and ability to stand side by side with farmer or processor, creativity and ingenuity was required. ACA has helped with forming plans to keep organic integrity with remote desk audits and other creative and practical methods consistent with the regulations ensuring that organic products meet the standards consumers expect.
CACS is awaiting release of Strengthening Organic Enforcement Rule. We have been eager for quite awhile. I expect there will be plenty for this subcommittee and for Board to dive into.
Ela: When rule comes out CACS will have a lot to do.
12:43 PM ET: Materials Subcommittee update
Dave Mortensen, Chairperson, gives the Materials Subcommittee (MS) update to the NOSB and stakeholders:
Highlights of Research Priorities 2020 (I got this one): Process by which we reach the research priorities is to discuss among board members and solicit input from stakeholders through public comments process and board meetings. Sometimes the board realizes research would inform the direction of policies. Received 27 comments on research priorities. Ecosystem service assessment has been identified by many stakeholders as important. Concern noted about the length of the research priority document. Urged to consider ways to prioritize these objectives. Need to think about the nature of the research as well as how it should be done. Folks would like to see expansion of discussion of soil health.
Subcommittee has been working on this with input from other subcommittees as needed.
Ela: NOSB chose to put the research priorities out as discussion document in the spring for votes in the fall to enable more stakeholder input.
Oakley commenting on marine materials: Received comments from a number of folks to look at all marine uses across all committees to look at the bigger picture. Ideally that would be feasible, but practically more difficult. If we were to categorize might pose some challenges, but will discuss further.
Ela: Commenters stated there is a need for comprehensive review and materials committee is the place to do that. It really is a difficult topic and look forward to what you’re going to do this year. This is a hugely complex subject.
12:55 PM ET: Lunch Break
[The NOSB will plan on reconvening at 2pm ET.]
2:00 PM ET: Crops Subcommittee update (paper pots)
Jesse Buie, Crops Subcommittee Chair, turns to Steve Ela for discussion of paper pots.
Ela: Lots of comments on paper pot proposal. We spent a lot of time on the terminology.
Summarizes the public comments received on paper pots: Number said should pass as it stands and others opposed. Some farmers waiting on a final decision before making the investment. We will send this back to subcommittee. There are a number of minor changes that prevent voting on it, but should have a vote next fall. Comments about seed tapes and other products to “including but not limited to.” Several comments that were worried other products wouldn’t be able to meet bio-based requirement. Unfortunately, didn’t hear from alternatives. In terms of terminology, paper is a synthetic fiber.

In terms of terminology, “paper” is a synthetic fiber. It also gets complicated because it’s a bio–based product but extremely not biodegradable. [For the phrasing] “primarily of cellulose paper and bio–based non–cellulose based….” We actually did mean two separate things. Our intent was that the fiber be cellulose based. Comments said we should spell out additives. It gets really difficult because different products use different adhesives. Can’t go to 100% cellulose base because hemp and cotton are not cellulose based.
If people submitted composition of pot that is known bio–based does that substitute for analysis (a number of certifiers were very worried). OPWC states these products need the strength of virgin paper. Another comment about how we know degrades into the soil. OPWC also wants prohibited colored or glossy inks. Will consider it. Also noted that 100% bio–based content might not be what looking for. Dilemma is if make annotation, and manufacturers move to hemp, how does annotation react to landscape? This is not an easy topic. We are committed to working on this.
Buie: What’s thinking on hemp now?
Ela: Manufacturers think in 3-5 years they can move in that direction.
Oakley: There is a 2017 supplemental TR on newspapers and other recycled materials that the new members might find helpful.
Wood: Found comments against virgin paper compelling. I also understand virgin paper is going to have strength recycled paper doesn’t have. Could you speak to this?
Ela: Difference in newspaper listing and what working on here. Quality of recycled paper matters. Manufacturers said couldn’t really make pot that would hold up without virgin paper. Study from University of Maine indicated the same.
Oakley: Paper chain plots is as a transplanting tray and when put in the field. Can argue it’s recycled once put in the field. It’s complicated.
Jeffery: Wondering if testing in regulatory framework is precedent setting. Is this a radical change?
Ela: any time reference an outside standard we take the chance we’re referencing an old standard. Need to look at the wording in case standard changes. While it is procedure oriented, at some point we have to be clear and technical.
Ela moves to send petition back to subcommittee. Mortensen seconds.
- Motion succeeds: 15 yes, 0 no, 0 abstentions
Tucker: I want to clarify. From program’s perspective we will continue to allow paper pots during this deliberation period. We sent a letter to certifiers indicating this and asked that certifiers make decisions in good faith.
[Cornucopia supports the listing of paper pots because they are essential tools for many small soil-based farmers. However, Cornucopia argues that the notation should not be so broad as to allow all possible uses. We suggested that the notation include the following requirements to avoid abuse and encourage improvements in the industry in our written comments.]
2:28 PM ET: Crops Subcommittee update (cont. Liquid Fish Products)
The crops subcommittee turns to the discussion document on Wild, native fish for liquid fish products.

Oakley: This came out of the sunset review of liquid fish products in 2018. AT that time, there was indication that there may be products harvesting whole native fish being used exclusively for fertilizer. The TR is extensive and says there are fish harvested for meal, oil, and solubles—but not exclusively for fertilizer. A majority of materials OMRI listed include fish waste, by-catch, and mortalitys, which may account for the idea that fish are harvested for fertilizer. The wet reduction process is that whole fish are cooked and pressed. Solids and liquids are separated. Solids are meal. Solubles and oil come from liquids. Reached out to community for input, including possible annotations. Some producers think they are using products from byproducts. Additionally, using the waste products may encourage this practice. There could be issues internationally where oversight is less strong.
Bradman: Expands on concept from public comments that use of waste to make liquid fish products that could make harvesting fish for a given purpose more profitable. The original harvesting may not be as viable without the production of fertilizer from waste products.
Oakley: That is the initial issue the CS had with this material. Some comments noted we should not degrade the marine ecosystem to enrich the agro-ecosystem.
Romero Briones: This has a lot of implication for Indigenous communities dependent on wild fish stocks. It is troubling that we would utilize wild fish stocks for commercial purposes. The salmon have created all fertile grounds in California, for instance. The organic community should not only support that system but ensure that connection remains. Wild fish stocks as commodity could create opportunities for exploitation. There must be safeguards to ensure there is not exploitation. I know this is complicated. Appreciates the conversation.
Oakley: These points were also brought up by some public commenters. A lot of these fish being harvested are food for wild fish that feed communities.

Ela: The TR shows that fish are not being harvested exclusively for solubles, but there are fish harvested solely for the agricultural market. Is there harm in putting in an annotation?
Oakley: Grappling with this herself. Inclination is to explore the feasibility of an annotation to restrict exclusive use of meal, oil, and solubles. This use of fish does give a revenue stream for the solubles that would otherwise not exist. The TR is the definitive word on this issue. The meal, oil, and soluble industry also has a livestock feed use.
Mortenson: The fate of fish is more complicated than I understood with respect to harvesting. I was fairly certain we were harvesting fish solely for fertilizer, but that is not what we found. Likes Emily’s plan for an annotation. From an ecological point of view, understanding what species we are harvesting and the impacts of such is important.
2:47 PM ET: Crops Subcommittee update (cont. Biodegradeable Biobased Mulch)
The crops subcommittee turns to the discussion document on Biodegradeable Biobased Mulch (BBM) Film Annotation Change.

Bradham: Another complex topic. As you know we put out a discussion document. I’ll provide a summary of comments and talk about future plans. BBM is a plastic material that is an alternative to polyethylene used primarily for weed control (and also used for pest control). [Polyethylene is the most popular plastic in the world.] There is a lot of ambivalence in community.
Beyond Pesticides made a comment that synthetic mulches should not replace organic mulch.
The flip side is petroleum products are used heavily in organic production. I know decades long farmers who rely on these materials. There is a trade-off, and the concern that we are still using a lot of petroleum products. Concern is if we’re letting it [degrade] into the soil. There are also some concerns that degradable plastics are produced from GMO. There is a related comment that we should not be regulating the end-product.
The crux of the challenges with this material is we don’t know extent to which it degrades. The NOP has funded research but there are still outstanding concerns about whether biodegradable mulch fully degrades (depending on climate). There are concerns residues may be left in soil and could end up in waterways and introduce new source of plastics in the environment.
There are interesting comments on where to go from here. Number of commenters noted we should look at paper pots 85% content for bio–based films and step away from 100% biologically sourced material. There is a lot of frustration that the material is listed yet there is no product that can fulfill that listing. Concern that we don’t allow some fraction of bio–based content, then should remove it from National List. Going forward, many comments implore the Board and program to allow use of these materials. There is such demand for use of films in organic production.
Even if we allow some synthetically derived content, we are allowing the lesser of two evils. OPWC recommended a phased approach. They do support allowing this material and changing annotation while posturing an incremental approach with a move toward fully bio-based content. This would mean an annotation with minimum bio–based content. Another question asked about incorporating nutrients and pesticides in these materials. The answer was yes, but first question is how we as a Board move forward. Do we want to allow use of these materials and not require 100% bio-based or do we want to take it off the National List since there are no products that fulfill the requirement?
Jeffery: Could you unpack what you said about GMOs and fibers?
Bradman: There are concerns in the annotation that limits the use of feedstocks that are derived from excluded methods. One comment suggested that rather than disallowing feedstocks, the final product must not contain derived from excluded methods. GMOs are used to produce some petroleum products that become biodegradable component of films. The key issue is not the production method, but the end product. Some biodegradable plastic materials that are components of the film are derived using GMO or using other excluded methods.
Jeffery: If GMO corn stalks used, then that’s less of a concern?
Bradman: Not talking about these products being derived from conventional corn—that would present another host of problems. Rather, there is evidence that GMOs are being used to convert the petroleum base to a biodegradable material.
Jeffery: You can have genetically modified organisms [creating a product without GE materials].
Mortensen: Agree with what Mindee and Asa said. My recollection of presenters was that there are microbial populations that work on the carbon substrate. The answer we got from presenters was crop feedstocks would be used and there is little control over feedstock and most was likely genetically modified.
Ela: We allow non-recyclable biodegradable mulch, but comes back to whether getting full biodegradation in the soil. Do you think we should just have certifiers verify not seeing plastic fragments in the soil? How should we move forward?
Bradman: OPWC and NOC presented some ways forward. We could have certifiers working with producers. There will be a learning curve. We could suggest removal and composting in a certified facility, for instance. If used every year, it’s going to be hard to tell if left over from previous year. There are approaches we can propose.
Ela: Some concern would degrade enough it wouldn’t be easy to remove, but maybe we can require certain percentage be removed.
Oakley: Plastic that degrades in the soil is especially challenging. Might not be feasible to remove it when the season is over and it might depend on the environment. Many farmers may not know what a low percentage of bio–based content it has.
Bradman: In some products it has increased and there is a movement toward more bio–based materials. When look at strawberry production, it’s still based on plastic culture. Plastic removed from the farm, but put back into earth in a different location like a landfill. It is going into a bigger waste dump that will persist for years and decades.
Oakley: Wholeheartedly agree. These materials are complicated.
3:14 PM ET: Crop Subcommittee: 2022 Sunset substances review
Soap-based algicide/demossers:
Greenwood: There were no comments related to de-listing this product and a handful in support.
Ammonium carbonate:
Mortensen: Material used as bait alone or in combination with yeast. Ammonium carbonate is often used in systems of poultry and livestock management. Strong consensus for relisting.
Soaps, insecticidal:
Greenwood: No one recommended de-listing and certifying agents note that there are over 100 users of this product.
Vitamin D3:
Turner: Listed for use as rodenticide. A dozen written comments weighted toward relisting or neutral on the input. The material was noted as fairly ineffective, but over 100 growers use it as part of their strategy. Beyond Pesticides noted the death is quite painful for rodents and there is some toxicity for non-target animals.
Aquatic plant extracts:
Oakley: Material prompted the marine materials discussion. Large number of comments from producers and certifiers. It is widely used across crops, regions, and scales. Growers have used this material prior to the NOP’s existence. Small amounts are used by a large number of growers. Several manufacturers say the non-synthetic forms require more biomass for the same amount of product. Others note that an alternative exists and this should sunset. Should continue to look into environmental impacts.
Ela: The board split in 2015 regarding relisting this material. The micronutrients they contain are reportedly hard to find elsewhere. Do you have any comments on how to move forward on this diverse topic?
Oakley: Seeking to move forward in the fall with an annotation. Taking into consideration aggregating all marine materials, but it would be harder to make discreet recommendations for each material. This is what led to the TR in 2016. Looked at nomenclature and environmental impact at that time, but it was challenging. Crafted a draft recommendation from the fall panel on marine materials. We need to explore the economic impacts. Seeking feedback of marine experts on this recommendation to build scientific agreement before bringing it forward.
Ela: Thank you for working on this very difficult topic over your tenure.
Oakley: I am hoping to move this forward for relisting with an annotation. In response to the overall impact we have on our environment, one commenter said, “Don’t let the perfect be the enemy of the good.” We have to move this forward in a manageable, enforceable, and actionable way.
Lignin sulfonate:
Greenwood: No comments from public or on the webinar. Certifiers mentioned they have over 200 users so pretty strong support to relist.
Sodium silicate:
Ela: This material was controversial in last review. It’s used to float pears out of dump tanks. Received a couple comments in favor of it. While there are mechanical ways to get pears out, it’s expensive for small processors.
For sodium silicate, Beyond Pesticides said it should be de-listed.
EPA List 4 – Inerts of minimal concern

Bradman: Declared bias: I think all ingredients in pesticides should be publicly available and there should be no trade secret ingredients. I think that should be required in conventional and organic. Universal dissatisfaction with current situation. We reference List 4 in the National List, a list that is no longer updated. The EPA list includes substances that do not comply with OFPA, like NPEs (Nonylphenol Ethoxylates). We’ve had recommendations from Board and have foundation for how to move forward. Hope NOP will establish agenda item to reformulate plans to work with other agencies. NOC, Beyond Pesticides and many other organizations provide a detailed roadmap on how to move ahead. NOP should work with Safer Choice program to develop a list of materials that are acceptable for pesticide formulations and meet OFPA. This puts requirements on EPA. Issue raised was where refer to unmaintained EPA list is if stifling innovation. Answer is yes. There are products out there that aren’t being developed and they might meet concerns under OFPA. Current broken system is stifling product innovation. A number of concerns that if we were to recommend delisting this would create turmoil in the market and lead to reduction of materials and increase cost of many materials essential for organic production. For this reason, some groups argue for the relist.
If take proposals to work with EPA and develop a new list it’s likely many materials on list 4 would also be on new list. In many cases formulations would not change. Key here is given scale of our industry and importance of materials to control pests in less toxic way, we need a system that works. Biological Products Industry Alliance (BPIA) and related stakeholders say changing process would be extremely disruptive. One of concerns is a lot of regulatory work would be required and at least a ten–year process. One of the recommendations from BPIA was to keep List 4 and allow EPA inerts that have tolerance exemption. Do we approve relisting of List 4 or delist? Although not immediate, removal might get program started.
Tucker: we are aware of the problematic nature of this very outdated reference. A lot of thought was put into this a few years ago. At the time we talked about rulemaking it would have been significant economically and decided other priorities to be advanced. Landscape has shifted a bit and it might be ripe for rulemaking. I’m intently aware of the issue.
Bradman: OMRI notes that there is minimum of 3 brand named products that didn’t appear on List 4. Here are 3 products that could be valuable and would have economic benefit but are not available. Costs in restructuring and also a cost if don’t do anything.
Tucker: All economic costs considered.
Bradman: Pesticides and organic can go together. To extent we can support the pesticide industry to stay true to organic philosophy, it is important.
Ela: Not a simple procedure. Suggestions on how to proceed?
Tucker: OFPA does not allow us to add synthetic materials to the National List without a Board recommendation. If you vote to delist this, you need to propose something else. It would need to be a recommendation to remove but to add this other thing. It does need to reflect that complexity.
Ela: That helps give us ideas.
Rice: National List manager could help on how to move forward. We are often faced with complex issues, but this is ideal case to draw on program’s strength and the expertise of other agencies.
Bradman: Even in public comments there was stated the need for a list manager. We need to come up with timeline to move forward. Concerns about staffing at EPA. I wonder if USDA could help EPA designate a staff member as a liaison to operationalize some of the road map that is needed. These things cost money, but if look at scale of pesticide market, this is situation where need to spend the money to strengthen integrity around pesticide development.
Ela: We won’t work on this in a vacuum. Concern when have a number of materials that shouldn’t be in organics, we need a long phase-in time to transform and re-register. We have to proceed without shooting selves in foot.
A-dae Romero-Briones: NOSB has to be diligent about process. I understand need for cooperation. EPA or federal agencies have their own political pressures. A lot of EPA regulations are put on hold at times. This warrants some discussion with NOSB on power dynamics and how powerful or not our recommendations. We have to consider political rollbacks in the future.
Bradman: Our standards should be very high and if work with EPA want those high standards be incorporated in any relationship and how we evaluate inerts.
Arsenic:
Buie: All seven comments received do not want arsenic removed from the list of prohibited substances.
Strychnine:
Buie: All comments received do not want strychnine removed from the list of prohibited substances.
The meeting will reconvene at 11 am ET tomorrow, April 30.
Thursday, April 23, 2020: Public Comments
[Public comments were given exclusively via webinar this year due to response to the COVID-19 pandemic.]
Dr. Jennifer Tucker, NOP Deputy Administrator, welcomes the NOSB and thanks commenters and audience.
NOSB member attendance:
Organic Producers
Jesse Buie (January 2016 – January 2021)
Emily Oakley (January 2016 – January 2021)
Steve Ela (January 2017 – January 2022)
Nathan Powell-Palm (January 2020 – January 2025) – new member
Environmentalists / Resource Conservationists
Asa Bradman (January 2017 – January 2022)
James R. “Rick” Greenwood (May 2018 – January 2023) – absent
Wood Turner (January 2020 – January 2025) – new member
Consumer / Public Interest Advocates
Dan Seitz (January 2016 – January 2021)
A-dae Romero-Briones (January 2016 – January 2021)
Sue Baird (January 2017 – January 2022)
Handlers / Processors
Kimberly Huseman (January 2020 – January 2025) – new member
Gerard D’Amore (January 2020 – January 2025) – new member
Retailer
Mindee Jeffery (January 2020 – January 2025) – new member
Scientist (Toxicology, Ecology, or Biochemistry)
Dave Mortensen (January 2017 – January 2022)
USDA Accredited Certifying Agent
Scott Rice (January 2016 – January 2021) – technical difficulties/not present at start of webinar
Public comments:
Kyla Smith, Pennsylvania Certified Organic (PCO)
Skipped/not present
Maddie Kempner, NOFA‐VT/VOF, Policy Director
Proposal for paper planting aids: thanks the NOP for extending allowance for use. Paper pots save time and labor for small organic vegetable producers. Adhesives and synthetic fibers are currently in other allowed papers and so should be allowed in this case.
Biodegradable bio-based mulch (BBM) should be allowed, but the 85% requirement would mean this material is not currently achievable. Seed tapes and other materials need to remain available.
Phthalates in dairy milking equipment need to be researched—there are alternatives and known human health effects.
Ela: Would things like seed tapes not be allowed under the criteria we mentioned?
A: Can follow up with the NOSB on that.
Robin Hadlock Seeley, School of Marine Science and Ocean Engineering, University of New Hampshire, no current academic affiliation; retired from Cornell.
Thank you for organizing marine panel. NOSB and NOP need guiding principles (1) do no harm; (2) end fragmented review process and hold to wild crop standards
Supports Beyond Pesticides’ comment on fish oil. Supports delisting kelp to require it be produced organically. Supports NOC position on liquid fish products.

Oakley: Any specific suggestions from panel we should consider as we develop standards and consider annotation?
A: (1) cautionary approach given the rates of ocean change; (2) putting protected areas off limits to harvesting
Bradman: What if we have a source that is only by-catch
A: That’s why I’m advocating a holistic approach. Fishery regulations may not account for by-catch. Calling it by-catch doesn’t account for everything.
Harry Rice, Global Organization for EPA and DHA Omega‐3s (GOED)
GOED’s mission is to ensure that members produce products consumers can trust. Fish oil annotation: GOED supports sustainable fishing; supports changing fish oil annotation, but there are concerns about references to FAO and NOAA issues. Some information on sustainability is too old to be accurate. Requiring third party certification would address environmental concerns and would not create more of a burden. Modify annotation to include: sourced from fishing industry byproduct and certified as sustainable from third party certifier.
Wood: Restate percentage of salmon and menhaden consumed by humans
A: 1% of salmon and 2% of menhaden is consumed by humans from catch in US.
Bradman: Relying on third party certifications external to the NOP is problematic because those standards can change and may not be reliable in terms of implementation. Why can’t these fats come from plant sources?
A: Different products. It is difficult to figure out which third party certifiers. GOED uses the most reliable in the industry.
Michael Crotser, CROPP Cooperative, Certification Manager
Sunset review of whey protein concentrate (WPC): supports removing from 205.606. CROPP produces whey. Organic WPC is fully available and supply will grow. CROPP’s supply is greater than market demand and processing infrastructure is in place.
We use fish oil, including 205.606–listed fish because there is no organic alternative. Three category system cannot access current sustainability. Utilizing third party standards can verify that production maintains ecosystems. Supports adding annotation that fish oil is sourced from fishing industry byproducts only.
Oakley: Marine Stewardship standard also looks at economic impact and social justice. Could NOSB look at this in annotation since it is beyond the scope?
A: They don’t directly relate to the standards but anytime there are additional attributes, we look at those on an economic and social level as a cooperative.
Bill Wolf, Wolf, DiMatteo + Associates – Founder and President
Earthworms are a guide to best practices: when considering what should be allowed in crop production, ask what encourages earthworms. They are good barometers.
We need our tool box. Renew listing of aquatic plant products, EPA list 4, vitamins, etc. Does not want anything dropped until there are replacements.
Apply commercial availability to the National List. Require organic “when available.” Prioritize which inputs most need to be reviewed for their impact.
Oakley: Elaborate on prohibition sodium nitrate?
A: Vote to remove sodium nitrate allowance, to align NOP standards with international standards. The vote occurred but then stopped with the NOP. The 5-year review has been ignored for sodium nitrate, still posted on 602.
Stephen Walker, MOSA Certified Organic – Accreditation and Industry Affairs Manager
World has changed and we need a new way forward. Organic research is the way forward. COVID reports note global disruptions. The pandemic offers a glimpse of the new normal, a fertile ground for a new food system. Research priorities can help us address climate change and protect the vulnerable. Now is the planting time for necessary seeds of change.
Oakley: Thanks for comment
Mortensen: Helpful to hear
Sam Welsch, OneCert, Inc.
Consistency and clarity: if it can be misunderstood it will be! The correct classification of malic acid—the synthetic vs. non-synthetic decision tree is not being used consistently. Some are considering the microbes to be the source substance. The microbial action is how the source substance is changed. The proposal for listing paper pots is an example of confusing language—paper is a complex substance. The listing needs to be revisited. Microplastics are accumulating and known to be detrimental to soil microbial matter. Research shows worms are smaller when there are microplastics in the soil.
Q: Recommendations for paper annotation?
A: Written with reference to other standards—it will make it difficult for certifiers to evaluate. Will offer further thoughts directly to the NOSB.
Joel Kelly, Live Local Organic
Certified organic farm using aquaponics. Chose to use container production because no other option for growing out of season in Portland’s climate. Last four months show need for food security. Supports aquatic plant extracts and revisiting container certification. Clarity is more important now than ever. If and when the issue is revisited, we need to consider public opinion. Experience has been strong public support for organic container production. Restrictions in container production is short-sighted, but further clarity is needed.
Baird: Do you differentiate between containers, roots in water, and aquaponics?
A: Difference in aquaponic and hydroponic production. Aquaponic production uses fish, so involves maintaining a biological ecosystem. There still is the issue of making sure there is a local and organic option, especially in climates with harsh winters. Certification should be protected.
Mortensen: As a Board member who has worked on this, we would need a stronger signal from the NOP that this is something we are working to resolve. Discussed on and off next two years and to have it more squarely on the work document; we need a stronger signal sent to the NOP.
A: If we can help with that, we would be happy to.
Seitz: Echo what Dave said. Appreciate you advocating for standards. Made a statement about consumer support for container production, but would you support labeling of container production so consumers can make an informed choice?
A: Absolutely. Thinks it’s a great solution so consumers can make an educated decision.
Patty Lovera, Organic Farmers Association
Top priority: maintaining integrity of label. The NOSB needs more ability to set their own work agenda.
Messages to the NOP:
- Need Origin of Livestock (OOL) Rule to address inconsistency on the ground. You can transition one time per producer. We need a quick effective date.
- Need focus on compliance with pasture rule, especially with high risk operations
- Need Strengthening Organic Enforcement Rule
In pandemic situation, farmers are acting quickly, but we still need USDA’s pandemic response to include organic farmers. Even though they are less consolidated than conventional producers and more adaptable, they still need help. Not clear that existing programs will work for organic farmers.
Tucker: Organic Enforcement is currently with the Office of Management and Budget; OOL is coming out of legal review (awaiting clearance of different departments)—both are moving forward. Pasture compliance project continues in its third year. Pandemic response: monitoring closely what is happening. Recently released fact sheet. The NOP is no longer committing to timelines, and will report instead on the process as rules advance through necessary departmental review.
Cali Alexander, H.O., NOFA‐NJ Board‐Policy
Skipped/Not present
Kestrel Burcham, JD, The Cornucopia Institute – Director of International Policy
Read Kestrel Burcham’s full testimony to the NOSB.
Mary Capehart, Organic Valley, Certification Senior Specialist
Commenting on biodegradable bio–based mulch: plastic mulch is nearly impossible to recycle and there is a large volume going to landfill. Find a solution to minimize plastic mulch.
[Technical difficulties; commenter hard to hear.]
Harold Austin, former member of the NOSB, handler seat – Chair of Northwest Horticultural Society
Crops list 4 inerts—this has received scrutiny and hard work, but we cannot de-list without something in its place. This is essential until an alternative process is implemented. Time to implement 2015 recommendation.
Aquatic plant extracts: these materials are used to compliment other organic practices like cover cropping, mulching, composting, etc. To improve soil fertility. Used when as-needed, when a problem /deficiency exists.
In handling, ozone is an important sanitizer. It is benign in the environment; used to help control microorganisms. Having ozone along with other materials is more important now than ever before.
Agrees with Bill Wolf’s comment: we should remove sodium nitrate.
Oakley: What should the NOSB do to move inerts issue forward?
A: Put back on workplan or have commitees draft language/resolution that action be taken.
Ela: Should we ask that sodium nitrate be prohibited?
A: Yes, it should be prohibited not just kept at 20%.
Harriet Behar, former member of the NOSB – certified organic farmer, educator, and inspector
Impressed by how many consumers write the board expressing their support of organic integrity. understand label is overseen by USDA. Encourages the USDA, as overseers of the organic label, to consider the long–term consequences of adding materials to the National List. The change in the voting procedure makes it hard to remove material from the list. Once a material is allowed in commerce, the NOP may decide to keep I there forever, like sodium nitrate, which has been on the National List for eight years without a single sunset review. It is effectively forever on national list, which is illegal.
Cornucopia note: The original purpose of the sunset process was to allow essential use of non-organic and synthetic materials in organic agriculture and food production for a period of five years. It would then take a two-thirds vote from the board to renew them for another five years. This was meant to encourage the development of organic alternatives. In 2013, the NOP unilaterally turned the process upside-down. It now requires a two-thirds vote to remove any material from the National List. This has de-incentivized the development of organic alternatives, as it is very difficult to gain 10 votes for a material’s removal, particularly given commercial interests increasingly represented there. Read more background on the noted changes to the sunset review voting process.
Fenbendazole: The fact that there is fenbendazole residue in the eggs is a rejection of the public trust. This material should not be allowed.
Biodegradable mulch – do not change annotation until it is shown to cause no harm
Whey Protein Concentrate needs to come off the National List.
Oakley: Requests the NOP’s comment on sodium nitrate.
Tucker: Sodium nitrate is not on regulatory agenda. The NOP sent a memo to certifiers on how to consider the listing, found in the NOP handbook. We will take this feedback back up to leadership but not currently on the work agenda.
Oakley: Given the confusion, there is some indication it is being used above the 20% limit. Is program aware?
Tucker: Not aware.
Seitz: Is fenbendazole necessary in poultry? Are there methods that would obviate the need for allowing it?
A: Rotation of pasture where you are aware of life cycle of parasite and not bringing birds back into the area where there are larvae for birds to consume. Some operations don’t have much outdoor access for birds, so don’t have sufficient space for rotation. Until we have a rule for 50% vegetative area for birds, there could be more rotative pasture. Has seen operations with 3,000-4,000 birds that rotate and they would meet the rule. Concerned that many do not have enough space. Allowing the parasiticide would encourage crowded conditions. This would encourage routine (non-emergency) use and would create constant residue in eggs–which consumers would find abhorrent.
Greenwood: Fenbendazole may end up as a residue in eggs, but it is destroyed at 179 degrees centigrade. Since they are cooked, don’t think that’s a particularly important issue.
Cornucopia note: Consumers eating a raw diet seek organic eggs in part because they do not contain pesticides.
A: Some of birds might end up as slaughter, so that should also be considered.
Greenwood: but still going to be cooked. Just wanted to reiterate point.
Wood: Finds value in comments, including the perspective on procedural changes.
Mortensen: We are talking about approving a synthetic pesticide. Idea we would allow residue in organic eggs is disturbing and worrying to me. A cultural method could address the parasite problem from the systems point of view.
Vamshi Chintha Rana, Meal Solutions LLC
Skipped/Not present
Patrick Kerrigan, Organic Consumers Association
Replies to issue raised by Harriet: organic consumers would be horrified to hear there is a synthetic pesticide in their eggs; this would hurt organic integrity. It could receive widespread media attention.
Challenge with vulnerability in food system due to virus; opportunity to invest in regenerative organic. On our way to build ecological food systems. We need to bring new farmers in and keep current organic producers on the land throughout this crisis. NOSB should urge USDA leadership to protect organic farmers. The USDA can best support farmers as recommended by Beyond Pesticides: financial assistance, virtual platforms for sales, and other safe direct to consumer distribution channels. Farmers markets and others need to be classified as essential.
Organic dairy farmers are still in crisis.
Jorge Gomez
Skipped/Not present
Emily Musgrave, Driscoll’s Inc. – Organic Program Manager
Focus on biodegradable bio–based mulch (BBM): Use of plastic is alarming and has become integral. Growers and consumers are acutely aware of plastic footprint, and many growers have expressed desire to use BBM while cutting down on plastic mulch. Should rewrite language on BBM–it is clear organic growers want to reduce use of plastic.
Insecticidal soaps – supports continued listing
Aquatic plants extracts – supports continue listing; commonly use seaweed and kelp
Bradman: Curious about plastic in strawberry production, a plastic intensive crop. Would BBM replace that use? To what extent would that be adopted?
A: Know a lot of growers that want BBM.
Ela: Concerns BBM won’t fully degrade?
A: Something to think about. However, I see the plastic waste. We talked to 50 recyclers and no one would take the plastic–it is simply going to landfill. Something needs to be done in the meantime.
Ela: What if we required it be removed and composted?
A: I think that would be a good alternative. Maybe Lynn Coody recommended certifier reviewing ingredients. There are different avenues, but what you mentioned would be viable.
Oakley: Does it break down in hot, dry climates? Are growers using there?
A: Good point. Some of our growers are in hot, dry climates. We need to look into this and need more research so growers can use BBM.
Brian Baker, IFOAM North America
Asks that all farmers who meet the requirements of the global organic guarantee system have access to markets. NOSB made recommendations about grower group certificates, but the draft guidance has not been published and there is no opportunity for public comment. Asks that there are no unfair barriers to trade, especially in poorest regions. Certification and inspection should be risk-based. Certification requirements should be adapted to regions. Scale should be considered as risk factor.
Baird: About fenbendazole issue: (Brian Baker was the principle investigator on the TR when he worked at OMRI) the rule says we have to prevent contamination with organic product. Feeding fenbendazole to chicken, is the final product the chicken or egg? Are we preventing contamination if it then disintegrates? Is it just a processing aid?
A: I thought the petitions was for eggs and not meat birds. Different temperatures at which eggs are prepared than what’s baked in oven or fried. Fenbendazole has only recently been labeled for use in poultry. When it was off-label use there was a zero-tolerance standard.
Dennis Dean, U.S. Citizen, consumer
Skipped/Not present
Ki Song Lee, ISC Agricultural Development Research Institute
Skipped/Not present
Kate Mendenhall, Organic Farmers Association – Director and organic farmer
OFA members top concerns: (1) NOP enforcement (2) organic import fraud (3) prohibit hydroponic production (4) climate change (5) dairy crisis
Healthy soil is crucial. Since NOP declared hydroponic production is allowed, certifiers have been creating own standards which is undercutting label.
Agrees paper pots should be allowable synthetic. Helps small organic farmers.
Does not have position on biodegradable mulch. With domestic and import fraud and three-year transition for hydroponic operations being determined on per certifier basis, bio-based mulch seems low priority.
Jennie Landry, DSM Nutritional Products
Manufactures Omega-3/EPA/DHA products. Suggested fish oil annotation change: source from industry byproduct only and certification by third party certifier should be added to requirements. Recommend third party sustainability certifications because it is clear and enforceable and not burdensome. Concerned about NOSB classifications from FAO and NOAA as fish oil for consumption is irrelevant to these classifications. It would cause confusion and be difficult to enforce if this is included. Also these requirements would potentially reduce fish oil availability.
George Seaver, Ocean Organics Corp
Has been making rockweed fertilizer for over 40 years and supports relisting aquatic plant extracts. Sustainable harvest is managed by state governments. Essential fertility tool and quantity used per acre is miniscule. Previously provided documentation about Maine’s oversight. Coast of Maine is about 3,000 miles, and only a fraction offers commercial harvest. No studies show damage. No reason to think ecosystem will be impacted with responsible harvest. Total harvest from Maine waters has grown slowly. As long as the Department of Marine Resources oversees, it should be allowed in organic food.
Dave Chapman, Real Organic Project/Long Wind Farm
Grows greenhouse tomatoes in fertile soil in Vermont. Wants to introduce new NOSB members to challenges in organic community. The NOP is going further from organic goals by allowing the spraying of hydroponic greenhouses yearly with prohibited substances. The NOP has failed to enact NOSB recommendations despite the recommendations not being radical. There are currently three lawsuits against USDA concerning integrity, dealing with hydroponic production and animal welfare. According to the NOP, these regulations would lead to the de-certification of the majority of egg operations and vegetables produced hydroponically.
If we accept that organic does not really stand for healthy soil and allow hydroponic production to be certified, the organic label will die.
Seitz: Earlier person said if we were to remove org certification of container production, then people in that industry would be in unfair competition with conventional hydroponic producers. Is there a competitive disadvantage with being soil–based compared to organic hydroponic production?
A: The major organic hydroponic producers are ALSO conventional producers. It’s all one and the same industry, just a matter of what they put in the nutrient tank. Hydroponic production is cheaper than soil-based; costs less to produce. That’s why they can afford to charge less. If that is the only metric to decide what should be certified, then essentially we are going to mandate it. It has become the norm for some crops, like tomatoes. It’s not possible to compete unless there is a distinction made in the marketplace.
Baird: Rotation is an important part of organic, not sure how we rotate hydroponic production. People focus on soil but I see less issue with soil as an issue with biodiversity.
A: Hydroponic producers inherently have less biodiversity. Coco coir has less biodiversity than the same volume of soil.
Wood: Do you have a position on moving forward with certified hydroponic production if you labeled hydroponic as such?
A: I cannot support it because hydroponic production is not organic, but it would be better than what we have now. My dream would be the hydroponic producers create their own bio-ponic label. I think they would have a market for that.
Pam Krone, California Marine Sanctuary Foundation
Under contract to Monterey bay Marine Sanctuary. Main concern is to have agriculture help and preserve ocean health. Microplastic is found throughout the ocean, including in Monterey sanctuary, throughout water column allowing plastic to be ingested throughout food web. Microplastic density in agricultural streams is comparable to Monterey Bay. Agriculture is contributing to ocean plastic problem! The most common source is plastic mulch. The PE mulch can’t be recycled. The only current way to get rid of plastic mulch is at a waste facility—about 10% gets left in field. BDM could be a sustainable alternative with the same benefits. The material cost is higher but the labor costs are reduced.
An organic certified biodegradable mulch is needed in the US. Consider adopting progressive standards for organic.
Bradman: What about plastic use on hoop houses and high tunnels?
A: Investigating different products that can be recycled. I’m not sure who, if anyone, is recycling these products.
Oakley: A large percentage of agriculture in that area of Monterey is conventional. Should the board consider not allowing plastic mulch at all?
A: There are so many benefits of BDM. Not sure what the economic issues would be and whether organic could survive. Would hate to see organic strawberries go away.
Jeffery: Referenced study in one of the Cal State schools—could you provide more info from that study? (Board notes they will reach out).
A: We have not written formal report for the study; we just finished data collection.
Ela: Do you worry about BDM not breaking down all the way? Wouldn’t that create the same problem?
A: Interesting to find out how long it takes tBDM to break down. Weathering does break down conventional plastic, but it’s very slow. BDM would break down much faster.
Anne Ross, JD, The Cornucopia Institute – Director of International Policy
Read Anne Ross’ full testimony to the NOSB.
Annie Kustermann, Beck’s Hybrids
Skipped
Javier Zamora, JSM Organics, Inc
Independently owned label. Small farmer and immigrant feeding families and communities. Keep in mind some NOSB decisions aren’t the best for smaller farms, including allowing hydroponic production. Small farmers lack a voice in the process, although we have major health, economic, and mental impacts in our communities.
Ela: One thing board could do to help you?
A: Marketing and insurance are big issues for smaller diversified growers. Large farms, including corn and soy farms, can get whole revenue insurance easily.
Greenwood: How many acres do you consider a “small farm”?
A: Small organic growers in our area have no more than 20 acres. I started really small and am up to over 100 acres. Once you start getting into 100s of employees, I don’t think it’s small anymore.
Gwendolyn Wyard, Organic Trade Association – Vice President of Regulatory and Technical Affairs
Regarding the question of whether malic acid should be classified synthetic: If the starting material or source is petroleum it would defy logic to end up with a natural substance. In our comments we’ve offered some clear allowance for a synthetic form when non-synthetic is not available.
Ion exchange filtration (IEF): It is important to understand this allowance is from 2002 NOP policy and guidance from 2008 and 2010. Certifiers are now reviewing these issues and whether ion exchange resins need to be petitioned to be added to the National List.
Rice: Question in discussion doc: addresses impacts of how we classify this on other substances. Classification of this as a synthetic might impact other substances on 605(a).
A: We analyzed other materials (notes their written comments) and as long as you view two-step production of malic acid as one process and focus on starting material of first step then there shouldn’t be an impact on other material listings. In no other example is the final product a petroleum product. The issue is what constitutes a “source material” for substances produced via fermentation. Substrate probably will include multiple inputs, which complicates things.
Eli Chandler, Thorvin, Inc.
Thorvin provides kelp ingredients for human and animal uses. There are no direct replacement for aquatic plant extracts, and they are a very efficient use of natural resources. The harvest of parent materials is regulated by government agencies. We support aquatic plant extracts being relisted for use in organic systems.
Kelp should not be relisted. There are sufficient quantities for animal needs and human uses. There regulatory definitions vary between animal feed and human uses.
Brad Johnson, Ingredion – Senior Manager of Process Engineering
To improve safety, quality, and purity of products they produce, we use ion exchange. Resins that are approved capture ions to leave demineralized product. The resin can only operate for a limited time since it has limited capacity. Acidic or basic solutions must be used for cations and anions respectively to remove waste. Then there is a rinsing step (pure water).
Ion exchange purification does not alter syrup being purified, only unwanted material is removed. Neither NaOH or HCl remains in the product or is in contact with product.
Greenwood: How is the waste processed and where does it end up?
A: Chemical and impurities are liquid waste. Water treatment facilities treat liquid waste before putting it back in waterways.
Angela Schriver, Shriver Organics, LLC
Grows row crops in Ohio, member of Organic Farmers Association. Wants to address domestic and import fraud. Her farm entered the organic grain market and spent three years transitioning to organic. Although confident in decision, we are still waiting for a rule to address fraud. Slow action or inaction on the part of the NOP is hurting our bottom line, livelihood, and family. There are enough challenges out there. Don’t make us wait for the rule.
The Board should not consider viability or scalability of farms when making decisions.
Poultry welfare: agrees unethical to withhold treatment if farmers need fenbendazole. Those treated can then be separated and not marked organic.
Oakley: Thanks for taking time to provide this testimony.
Ela: Fenbendazole usually used to treat whole flock. Should all be removed from organic?
A: Once you scale up to a certain size, then that’s a risk the farmer is willing to take. Consistently need to find other ways to manage animal health. Yes, I believe if treated, all should be removed from organic.
Meagan Collins, Accredited Certifiers Association, Inc. – Coordinator
Do certifiers and material review companies need to review cellulose component, and does this include all types of cellulose? “Primarily cellulose based” is unclear. We support paper planting aids but clarification is needed.
We have been working on best practices when in-person inspection is not possible due to COVID. Trying to address pre-competitive problem. Stresses importance of in-person NOSB meetings when able.
Ela: Can you recommend what would make it more clear?
A: Giving percentages we could verify cellulose at would be helpful.
Mark Hutton, University of Maine – Vegetable Specialist and Associate Dean
Wants to update NOSB about 100% bio–based paper mulch. Over past 10 years we’ve been working to develop paper mulch as an alternative to plastic. Many engineering obstacles: thickness, weight, capacity, and durability. Materials are 100% bio–based and the raw materials are sourced from forestry suppliers. Concerns around virgin pulp fiber quality, impurities, etc. in paper mulch.
Bradman: In terms of water conservation, how does it compare to plastic? Are there other fiber sources that are non–forest sources to prevent cutting down trees?
A: Paper mulch does conserve moisture. Watering frequency is no different from plastic mulch. Impact on forest production would not be very significant. One of the companies we work with could produce enough mulch in three days to replace all plastic mulch used in the marketplace for a year. We haven’t looked at other pulp sources but could be a possibility.
Oakley: What are biobased coatings?
A: Nanocellulose and sucrose esters
Wood: Do you have data on cost comparison?
A: As we have been designing this, we have been trying to hit a price point. Pick up and disposal costs run about $350/acre for plastic and we think we’re in range. The product will cost more than plastic, but trying not to exceed $350/acre.
Jeffery: Would you be able to provide more info on nanocellulose?
A: It’s tightly bound pieces of carbon. Slow biodegradation.
Ela: How does it hold up if stepped on?
A: Not well.
Ela: Talking about all virgin pulp or including recycled paper?
A: Sourcing recycled pulp is challenging.
Sarah Alexander, Maine Organic Farmers and Gardeners Association – Executive Director
Without consumer trust, organic will become another confusing and meaningless label in the marketplace. Prioritize rules on organic enforcement and origin of livestock (OOL). Please do not leave loopholes in OOL that would allow the problem to continue. Urges NOP to continue pasture rule enforcement, focusing on 1,000+ cow operations and those on margin of DMI allowance.
NOP is allowing hydroponic operations without any clarity on how they fit into the label. Calls for moratorium on hydroponic operations until there is more clarity.
Ask that they continue in-person meetings; they provide transparency that is important.
Ramzi Sulieman, True Organic Products, Inc – Chief Operating Officer
Speaking about the importance of liquid fish as a fertilizer. True Organic Products is leading producer of fertilizer. Byproduct of fish oil and fish meal is essential for liquid fertilizer program. As indicated in TR, byproduct would be wasted or thrown away. Products we use capture waste. We support findings of TR that none are harvested exclusively for fertilizer use.
Jeffery: Can you speak about different properties in byproducts of fish versus the whole fish?
A: For the most part they are very similar. Some fish that don’t make it through any quality programs they send over for fish oil or fish meal, but most of the waste is from scraps. We haven’t seen much variation in fish.
Dave Shively, OEFFA Grain Growers Chapter
Farms in NW Lake Erie watershed, raising corn, soy, wheat, and cover crops. Past president of OEFFA. The last few years have brought decline in market price, especially in corn. There is an excess of corn in market due to imports. NOSB and NOP has set rules for high standards for his grain, but the imports do not have the same traceability. Imported “organic” grains should not be unloaded without testing at point of entry. There should be more pressure to close loopholes on these imports. The Farm Bill has dedicated funds for this. The longer this enforcement is delayed, the more organic farmers are hurt.
Could you please change dates for NOSB meetings to make it easier for farmers to attend?
Wood: Given the difference in planting times across the country, when is a good time to have NOSB meetings?
A: Earlier in year, February or March. And the fall is bad for people harvesting, so earlier in the summer there.
Baird: What procedures would you suggest we could put in place that are not already there that could eliminate fraud in grain?
A: Grain keeps changing hands; changes from whole to cracked. It’s very disturbing. And domestic conventional farmers transitioning over don’t always understand that these standards are important. I look at it as a special crop with integrity and traceability, not just another commodity.
Michael Jones
Small organic grain farmer from North Central Ohio; belongs to OEFFA Grain Growers Group; first certified in 1983. We all struggled back then to succeed as farmers, producers, and processors, and we wanted to produce high quality food. Sure, there were people back then who tried to cheat. Many organic farmers were willing to self-police to maintain the integrity of label.
In the last five years, prices dropped when the demand was going up. Fraud and lack of enforcement against it are to blame. Thankful for NOSB who have heard our concerns and passed on suggestions to the NOP and USDA. Need to continue to encourage USDA to finish the job and give us the opportunity to comment and strengthen the rule. The futures of my farm and others’ farms depend on it. Lost 30 to 35% of revenue due to imports. Greatest concern to be able to pass this farm on to grandchildren. Thank you for your work.
Kenneth Rider, Farm name: RIDER LANDING, Certifying Agency: OEFFA, Member of OHIO ORGANIC
Certified organic for 35 years. Certified by Real Organic Project. Organic grain cropping overview: there are many aspects that make growing grain very complicated (lists them). This intensity never stops for an organic farm family. We must have defined worldwide organic standards. We want to see updated evidence of these efforts. The penalties for fraud must be immediate—no offloading of ships. Please verify.
Baird: Thank you for walking us through that. You are the backbone of our organic industry.
Edward Andrews, Andrews Farms Inc.
Andrews Farms takes care to follow the rules. Other farmers don’t follow the rules. Some of the certifying agents don’t follow the rules. There are some real abuses in the organic livestock industry too. Struggling now because of low prices. I don’t care where it comes from, we need stricter enforcement. We aren’t going to have a market to sell to. I’m asking for stricter and faster enforcement. I’m asking for the best you can do.
DeEtta Bilek, general public and OFARM
Canceled
Michael Menes, True Organic Products, Inc. – VP of Food Safety
Ammonia extract/novel ammonia products will harm the integrity of organic. Petitioning to prohibit this novel fertilizing material. New ammonia fertilizers are not compatible with organic. Ammonia extract will harm integrity because: synthetic ammonia fertilizers are chemicals, do not foster a healthy soil system, have high environmental impact, and deteriorate soil health. Synthetic and natural ammonia are hard to tell the difference between, so there is great potential for fraud. Please expedite this petition and prohibit this material at the fall 2020 meeting.
Oakley: Some folks are allowing use you said (materials review organizations and certifiers). Could you elaborate?
A: Yes, there have been other applications and at least one materials review organization has approved an ammonia product. There are many in line. The name of product is Biostar Perfect Blends 00.
Ela: how is this being produced “organically”; I know the petition is being produced.
A: There are biological ways of making this chemical fertilizer, including capturing from fermentation or composting. There is a definition we are proposing in the petition.
Jeff Dean, OEFFA Grain Growers’ Chapter
Co-owner of organic farm certified for over 25 years. Grows multiple grain crops. This is the fourth or fifth time I’ve addressed the board. Appreciate it. I spoke in Colorado and Pennsylvania. The times of meetings are a problem. Spring and fall are the worst time for vegetable, dairy, and grain producers. There has to be a better way.
Organic fraud: It has been over five years and it doesn’t seem like it has gotten any better. We know where it’s coming from. We know it’s not organic. Nothing happens until a ship [of fraudulent grain] gets unloaded. We have got to stop it and decertify it. If I got caught doing fraud I’d be decertified. This is going to kill the organic label. We work so hard to do things correct and we get hammered. We have suffered over $4/bushel loss for corn. We need to take urgent action now to take care of this problem.
Wood: Are you saying organic corn is $4 bushel?
A: Price has dropped $4 bushel. Now big buyers are using imports against us. If they can’t get farmers to come down in price, they will import grain knowing it’s fraudulent. Buyers don’t care if paperwork is in order.
Elijah Dean, OEFFA Grain Growers’ Chapter – Current Pres of Grain Growers Group
We meet in the early spring and winter to share information and work together to become better farmers. Purposefully held in early spring and winter because that’s when all our members are together. The NOSB meeting times are difficult. It would be easier to move meetings a month or two earlier than they are now.
The organic economy/industry is based on growers voluntarily complying with stricter regulations and consumers voluntarily paying more. Based on trust and integrity during intermediate steps. This integrity is being attacked: both products and practices being proposed. Fraud and fraudulent imported grain is also an attack. Ask that you maintain strong practices. If folks say we need new products because organic is difficult, good, organic is supposed to be difficult. It’s supposed to have strict practices.
Oakley: We have spoken with NOP about changing meeting time. That conversation is taking place.
Wood: Thanks to everyone in OEFFA grain growing chapter for taking the time to comment.
Steven Turnow, Turnow Ventures Ltd
Import and domestic fraud is a hot topic in the Midwest. A global pandemic has forced us to ask if we want to rely on foreign food. This country has the capacity to grow good quality organic food and soybeans. Why are we spending so much time and money buying food grown halfway around the world? We need to keep farmers transitioning to organic.
Michael Hansen, Consumer Reports – Senior Scientist
Colors: consumers expect organic to be free of artificial products, so using conventional colors is misleading and problematic. Residues of pesticides are present on vegetables used for coloring when conventional. Many certified organic products contain certified organic colors. There should be enough alternatives. We urge the NOSB to remove these colors from the list.
IOE should be removed from the National List. This is not necessary for production and only useful for marketing claims. This is highly processed, uses synthetic aids.
The crops subcommittee should continue work on table of excluded methods.
Bradman: Of the colors, which specific colors do or do not have the option of organic source material? Are there some that are more egregious?
A: Just based on the pesticide data program, it’s a problem. There should be enough organic products and removing colors would encourage the growth of those crops and want to encourage a market demand for those colors.
Ela: Some public comments said it is not a problem of availability of crops but that the crop is picked at different time for processing into colors.
A: But the same would be true for conventional vs. organic. It can be done with organic. I suspect people are using conventional only because they can get it cheaper.
Ela: so we should be looking at those colors that have good alternatives.
John Hendrickson, Small Farm Works
Submitted petition re: paper pots. Wants 205.2 to extend paper pots. Use has grown significantly and these pots are environmentally friendly. Many farmers tell me they would drop certification if they couldn’t’ use paper pots.
ASTM testing to determine the amount of synthetic material in the product is not reliable for paper products made from wood pulp. Test results made from long lived trees cannot be trusted due to background levels of radioactivity from historic testing of atomic bombs. His company seeks to bring hemp-based paper pots to market, as this would not have the same testing problems. They plan to have them available for the next growing season.
Albert Straus, Straus Dairy
Enforcement clarity and consistency is needed. Disconnect between farmers, retailers, and consumers is feeding the crisis we are in. The video put up by NOP was excellent direction to increase enforcement concerning documentation from farmers. Definitions around grazing seasons still need refinement for consistency. There needs to be consistency between inspectors, farmers, and consumers; everyone needs to be on the same page. We have the opportunity to look at food and farming that is more regionally centric.
David Epstein, Northwest Horticultural Council (NHC)
The Pacific Northwest produces the majority of apples, pears, and sweet cherries in the nation. Controlling foodborne pathogens is a major challenge for packing houses. We need effective sanitizers and disinfectants. Concerned about cross contamination and lysteria, including reduced sensitivity to single sanitizers. Need to use multiple sanitizers for effective microbial control. NHC asks NOSB to consult with FDA before taking further action. Want organic production to continue to thrive in the Pacific Northwest.
Bradman: Are there specific sanitizers in pipeline you’re referring to? Existing options are available. Can you be specific?
A: Made this statement to be general. If you’re going to be considering materials, you need to consider the requirements we’re required to meet. This is to ask NOSB to be mindful of mandates and your consultation requirement with FDA.
Bill Denevan, Viva Tierra
Asking NOSB to allow a product that controls fireblight in apples and pears. They are working on formulations that would be okay for organic, as long as the active ingredient is allowed (kasugamycin). Since streptomycin was de-listed in 2014, 20 new organic blight controls have hit the market, and none of them have been very good. Eleven recent studies show that these products are not as effective as conventional kasugamycin. Climate change is causing warmer winters, erratic bloom, and blight that is worse.
There was a petition submitted recently for this material.
Alicia Bock, Agrisystems International
List 4 inerts: I agree with OTA’s comments. It would be very disruptive if no replacement system. EPA list seems great place to start. In favor of relisting. Materials have been vetted and there are no viable alternatives. Even if no comments, it doesn’t mean material is not needed. If removed prematurely, hurts organics. Toolbox has gotten smaller. This is not the time to reduce annotation material for organic processors. Sanitizers are also necessary for food workers.
Ela: If transition from List 4 to Safer Choice Program, how would we handle discrepancies?
A: How we left it a few years ago between agreement between EPA and USDA. Have to take them on a case by case basis.
Steve: If transition from List 4 to Safer Choice Program, how would handle discrepancies?
A: How we left it a few years ago between agreement between EPA and USDA. have to take them on a case by case basis. combined effort between the two a
Tuesday, April 21, 2020: Public Comments
Michelle Arsenault, Advisory Committee Specialist, welcomes participants. Transcripts of comments and meeting will be posted to the NOP website and available about two weeks after the meeting concludes.
Dr. Jennifer Tucker, NOP Deputy Administrator, welcomes the NOSB, including five new board members:
Organic Producers
Jesse Buie (January 2016 – January 2021)
Emily Oakley (January 2016 – January 2021)
Steve Ela (January 2017 – January 2022)
Nathan Powell-Palm (January 2020 – January 2025) – new member
Environmentalists / Resource Conservationists
Asa Bradman (January 2017 – January 2022)
James R. “Rick” Greenwood (May 2018 – January 2023)
Wood Turner (January 2020 – January 2025) – new member
Consumer / Public Interest Advocates
Dan Seitz (January 2016 – January 2021)
A-dae Romero-Briones (January 2016 – January 2021)
Sue Baird (January 2017 – January 2022)
Handlers / Processors
Kimberly Huseman (January 2020 – January 2025) – new member
Gerard D’Amore (January 2020 – January 2025) – new member
Retailer
Mindee Jeffery (January 2020 – January 2025) – new member
Scientist (Toxicology, Ecology, or Biochemistry)
Dave Mortensen (January 2017 – January 2022) – absent
USDA Accredited Certifying Agent
Scott Rice (January 2016 – January 2021)
Public comments:
Philip LaRocca, CCOF – Owner and winemaker at La Rocca Vineyards and Chair of CCOF. 47th year as organic farmer.
Support rule of 3 year transition after use of prohibited substance, including greenhouses. Emphasizes strengthening pasture rules and maintaining vigilance on import fraud. Thanks NOP, NOSB, and organic producers and consumers. Hope for a healthy world rests in production and consumption of organic food.
Oakley: Are you aware of any transplant or seed production that might be transitioning using same for organic and conventional, or shortly thereafter?
A: Heard taking place, but no evidence. None of CCOF members practicing that to his knowledge.
Ela: Support three year transition for greenhouses?
A: Yes. If jump in and put a greenhouse up and land had prohibited material — this shouldn’t take place. It isn’t fair and shouldn’t be able to use prohibited material.
Pete Nell, CCOF – CCOF represents over 4,000 organic operations
Novel ammonia products: encourages board to prioritize petition.
Aquatic plant extracts have been used for decades. CCOF is unaware of alternatives.
Liquid Fish Products: question remains whether annotation is warranted.
Lynn Coody, Organic Produce Wholesalers Coalition (OPWC)
Paper pots issue, specifically the source of fibers. Biodegradability standard was not included, but the listing includes the words “degrade into soil.” The suggested listing needs to be corrected with respect to inks.
Plastic mulch is an important tool and there is an interest in using biodegradable mulch; OPWC comments provide a road map on how. There is market interest for mulch films with higher degrees of biodegradable content.
Q: How do you address differing biodegradation based on humidity of environment?
A: [This is why we need more attention paid to biodegradability.]
Peggy Miars, OMRI
Inerts of minimal concern—the subcommittee stated the proposal. Between 2011-2020 OMRI approved inert increased by 93%. Encouraged a memorandum of understanding (MOU) between EPA and NOP.
Bradman: Are there any substances on EPA 4 right now that should be prohibited? In principle, all ingredients in any formulation should be publicly available. I am curious to know which materials are not on 25(b) list. Any user or material should know what’s in the material and what’s being used.
A: OMRI does not take positions on whether to allow a material, we take cues from NOP. I don’t know which substances are not on 25(b). OMRI does not take a position on whether ingredients are publicly available. EU does not pay attention to inerts.
Aimee Simpson, PCC Markets – Director of Advocacy
Unprecedented challenges last six weeks with Covid-19. PCC prioritizes organic and, when possible, local producers. Organic, local, and small have proven essential. When large scale systems are failing, organic and local are lifeline to the community. Given stresses of time, calls for relaxed regulations cannot be tolerated. Organic label must be as strong as can be.
Oakley: Thanks her for comments and agrees.
Jay Feldman, Beyond Pesticides – Executive Director, former member of NOSB.
Welcomes new members. The NOSB has a special responsibility to maintain integrity. Short–term market growth should not be preferred over long–term trust in label.
Synthetic substances: should be an assumption they are kept out. Sunset means a substance does not stay on the National List unless it meets requirements of OFPA. We need more pasturing of animals, to preserve the marine environment, to ensure rigorous inspection, and to protect farms and biodiversity. If we do not adhere to these, we will erode trust. What do we do when USDA does not listen to NOSB? NOSB needs to assert its authority. We need to pursue it [so it isn’t just a niche market] to [prevent environmental collapse].
Kiki Hubbard, Organic Seed Alliance – Director of Advocacy.
Wants to emphasize keeping seed at forefront of work plan: pleased proposals were passed on topic of updating organic seed regulations and guidance for planting stock requirement. Genetic integrity: happy to see three research priorities related to seed. Urgently need a task force to collect baseline data of seed to inform policies. NOSB also well positioned to recommend how best to move forward with testing. Wants to see topic of excluded methods back on agenda and encourages NOP to adopt NOSB’s recommendations.
Baird: Getting requests to help hemp producers. Any organic hemp seed? Is there a lot of CRISPR used for hemp seed?
A: Hemp is not a crop OSA follows. Unfortunately, doesn’t have answers at the ready.
Jen Berkebile, Pennsylvania Certified Organic (PCO) – Materials Program Manager.
PCO supports paper pots and other production aids. An alternative listing of “paper crop planting aids” is a good idea. Would seed tapes comply? This is a concern. But requiring testing (ASTM percentage requirement) will limit some aids because some manufacturers will be unwilling to do the testing—but that’s okay. Hopefully there will be 100% bio-based pots someday, but there are not now nor will there be anytime soon. PCO will collaborate with other certifiers on this issue moving forward.
Thanks for holding remote meeting. We encourage NOSB to hold in-person meetings whenever possible.
Q: Fenbendazole for “blackhead” in your written comments?
A: 138 poultry operations interested in fenbendazole, a subset requested it specifically for blackhead. Clients asking if they can use it for this use?
Q: Blackhead is not a worm.
A: If fenbendazole is added to the list, we’d have to check if blackhead is an allowed use within the context of annotation.
Q: Paper pots have to last longer than seed tapes, so assumption was that seed tapes would meet bio-based requirement. Given annotation is written now, would PCO support it?
A: Yes, PCO supports current annotation.
Amalie Lipstreu, Ohio Ecological Food and Farm Association (OEFFA) – Policy Director
Members of NOSB are aware of benefits of national system of agriculture and it’s to the latter you must devote significant attention. Large bureaucracies are often unable to respond to needs on the ground. There is an opportunity for organic to shine. Organic agriculture should help mitigate threat of climate change. Systems-based approach; organic requires accountability. Need to look at regulations regarding soil management and establishing clear standards for container systems. Please add the role of organic agriculture in climate change to your work agenda.
Oakley: Can you elaborate on lack of soil provisions?
A: Most certifiers feel they are doing a good job of checking in with producers. We need to have a firm foundation in showing practices are being followed. I know when NOC held some discussion groups, there was variation in what was and what was not being verified. Board could provide greater consistency in terms of soil building practices and addressing need for standards and restrictions on container systems.
Nicole Dehne, NOFA VT – Certification Director for Vermont organic farmers.
Biodegradable bio–based mulch (BDM): there is no product on the market that can meet the criteria (100% bio-based polymers). If we are willing to accept some synthetics in papers, we should do the same for BDM. We need more research on what remains when BDM biodegrades. BDM can help reduce plastic use. Use the proposal for paper planting aids as a guide and require users to use a higher percentage of bio-based materials when available. Fenbendazole: these farmers do not review parasiticides to work. Pasture rules for poultry are inconsistent—how can you have a pasture plan without pasture?
Jeffery: Perspective on the barrier for evaluating BDM this far back from GMOs in source material?
A: Annotation is stricter than in others because GMOs are not present in the final product. So the BBM annotation is not typical.
Oakley: Are there any BDM products on the market that could meet the 85% working suggestion you have?
A: 65% bio-based material is the last manufacturer spoken to—so it’s possible there isn’t one that would comply with that now. But 85% is a good compromise comparing to paper.
Jaydee Hanson, Center for Food Safety – Policy Director
CFS strongly supports NOC comments to strengthen organic agriculture during COVID pandemic. We note that USDA has the final rule on organic livestock due June 17, 2020 as mandated by Congress and we are hoping NOSB will push NOP to meet that rule.
NOSB should defend against allowing new genetic engineering (GE) technologies Must adhere to framework in 2016. NOSB should urge NOP to publish guidance document – disturbed NOP has not finalized recommendations. New GE techniques being introduced at rapid rate. NOSB and NOP must provide clarity on which are allowed and which are prohibited. Some GE companies have mischaracterized their techniques as traditional breeding.
Baird: Interesting comment that some of laying hens could end up as slaughter—concern that the parasiticides need a specific withholding time for slaughter stock AND eggs if fenbendazole is allowed.
A: There are many cases when laying hens do end up being slaughtered. In pandemic time, conventional hens used for vaccine development have been slaughtered. Other hens are slaughtered in commercial operations.
Julia Barton, Ohio Ecological Food and Farm Association (OEFFA)
Anxious for proposal on strengthening organic enforcement—much needed to address domestic and import fraud. Fenbendazole: our experience includes only one request for parasiticides in the past year and a half. Concerns about residues in eggs. The NOP should move forward with changes to the “emergency” definition.
The tacit certification and endorsement of greenhouses is a problem. We need container growing back on the agenda.
One meeting each year could be held in person and one remotely—remote meetings allow for broader participation.
Oakley: Could you elaborate on the poultry technical review (TR) you are requesting?
A: The info currently being used is based on mammals, and poultry are different. The TR if it were completed, it would give info needed to make decision on fenbendazole.
Baird: Were you aware we did request a TR for the laying hens? We delayed this decision until the results of that.
A: No, glad to hear it.
Oakley: Could the NOP respond to requests from stakeholders regarding container growing issue?
Tucker: We are continuing to listen to feedback on this issue and others.
Mark Kastel, OrganicEye – Director
Reality check for new members: success of organic movement was based on integrity. After the smoke clears, we could have organic food without farms. Majority of organic dairy cows are now coming from livestock factories creating an illusion of grazing. Law requires access to the outdoors. New animal welfare rule also allows factory conditions.
Imports: After years of stonewalling, the NOP recently announced 75% of certified operations have lost certification. Congress ordered NOP to consult with NOSB. NOP tells certifiers to confer with other certifiers. NOSB needs to assert its oversight on how law is carried out.
Steve Etka, National Organic Coalition
NOC is busy advocating for federal actions for folks impacted by pandemic. Congress passed three packages, the third of which went to farmers. Still seeking more details on how organic farmers will be impacted.
The pandemic should not delay action on two rules: Origin of livestock and organic enforcement rule should be published immediately. One of NOC’s top priorities: promoting role of organic in climate change solutions. It means being honest in areas where organic can do even better, closing loopholes. Put more teeth into regulations that require farmers to use soil-building practices. We hope that the pandemic will help us figure out ways to make our food system more resilient not just from pandemics but from climate change as well.
Abby Youngblood, National Organic Coalition – Executive Director
NOC represents diverse stakeholder groups in the organic community. In 2020, NOC has prioritized: ways organic can address climate change, organic fraud, and the organic dairy crisis. Asking NOSB to limit highly soluble nitrogen in organics. Other areas of critical work: uphold prohibition against GE based on definitions and framework put in place in 2016. Six GE techniques still need to be reviewed.
Regarding three year transitions: Lack of clarity for greenhouses. NOSB should ask NOP to provide clarity. NOC also believes hydroponic and container systems are inconsistent with principle of organic farming and the law.
Liquid Fish Products as fertilizer: NOC support work agenda item to make sure not harmful to marine environment. Harvesting wild caught fish for exclusive use as fertilizer should not be allowed. Fish Oil: proposed annotation is insufficient to address environmental concerns.
Oakley: Three-year transition: previous attempts to get clarity have been unsuccessful. How can we get clarity?
A: Looking across certifiers to see what’s consistent and believe there is not consistency. NOSB should look at areas of inconsistency and request clarity from NOP
Oakley: we don’t have access to inconsistency across certifiers. do you have suggestions on how to get access?
A: Might be a good conversation to have with ACA.
Q: Why is current annotation re: fish oil insufficient?
A: Better to ask Christy Badger. Fish oil is not a necessary organic food ingredient. Doesn’t meet necessity or essentiality requirements.
Bradman: How do we regulate contaminated food and ingredients, given everything has some contaminants. How do we set standards? How to approach is complicated. Re: harvesting wild fish for fertilizers. In some ways, we harvest seaweed for fertilizers. There is an interesting analogy there in terms of harvesting from ocean and setting standards.
A: NOC does support use of liquid fish fertilize but only as waste product.
Alan Lewis, Natural Grocers
Natural Grocers has 157 stores, all of which are certified organic handlers. Retail is still a weak link in organic compliance, between pesticides and co-mingling in supply chain. Calls into question whether we are really delivering organic products to consumers.
COVID/food chain crises call out concentrated production as a huge problem. Silver lining: consumers are now more aware of local production. CAFOs and hydroponics are not the “cheap food” solution promoted. Hydroponic has reduced the quality of organic production, particularly tomatoes. Time to promote why supporting farmers is so important. Innate response: people are looking toward their diet and a cleaner environment.
Genetic manipulation is running rampant behind science. Do not ignore this as GE products enter the environment. Push back.
Good story: Farmers previously saying organic certification was useless are now reaching out because they wish they had a trusted seal. Retailers are having significant shortages in organic produce due to broken supply chains.
Robert Rankin, International Food Additives Council – Executive Director
Supports relisting waxes, calcium phosphates, corn starch…all are safe and consistent with organic principles. The eighteen colors at 205.606 are all essential. Reiterates purple potato, various carrot and elder berry juice are not alternatives. Organic corn starch demand can’t be met by US production.
Bradman: Challenge is when do materials come off 205.606 (National List). Curious what your members are doing to increase organic supply and if a program within your organization.
A: Not a program within IFAC. Member companies may have these types of objectives. Color industry does appear to be actively pursuing a larger supply of organic, natural food colors, but there are issues related to timing and contracts set up, like getting the land to devote to color side of it. Large majority of organic fruits are used for consumption. Five years would be a good timeframe to allow companies to get sufficient organic supply together and mitigate cost impacts.
Ela: You ask to relist all colors. I’m concerned we keep hearing five years. How do we move this forward instead of being put off?
A: I can only tell you what I hear from IFAC member companies. I can dig into each individual color and timeframes before the fall NOSB meeting, but I can’t speak on behalf of all the companies today. That’s where commercial availability comes into play. Agrees to offer NOSB more detail around each individual color in the near future.
Cornucopia note: Unsurprisingly, IFAC supports the continued listing of all food additives. They did so five years ago as well.
Marcelo Girotto, MPI
Skipped, not on the webinar
Dave Carter, Crystal Springs Consulting, on behalf of Merck Animal Care
On behalf of Merck, worked to expand petition for fenbendazole. Approval for mammals (2012) was a step forward in “continuous improvement.” Fenbendazole is a resource to have available in poultry; less harmful than other parasiticides. Protecting the health of animals is part of good stewardship. Ensures health of birds without harming health of soil. Expresses concern about what constitutes an emergency; next speaker will address that.
Baird: Concerns about 2.4 ppm residues in the eggs—can you address? There are also concerns that there might be residues in slaughter animals.
A: Slaughter animals: valid consideration in terms of withdrawal time. In eggs, delay that question because next speaker will address that.
Baird: Several certifiers said that none of their operations have requested use of fenbendazole or any other parasiticide. They say that enough rotation will be sufficient to prevent parasites. We did not have that come up with mammals.
A: Environment is not static. You can’t always follow a set schedule; emergencies do arise. Growers need to have the resources to address that so their flocks don’t suffer.
Blayne Mozisek, Merck Animal Health ‐ Technical Services Veterinarian
Veterinarian servicing organic and conventional flocks. Showed four slides, including birds with outdoor access and heavy worm burden, roundworm inside an egg, and blackhead (a very serious disease with no cure). Fenbendazole does not control blackhead but does control the worm that is the vector. This is an animal welfare issue.
Emergency use and residue decisions should be based on data. My understanding is that a modified technique can measure worm eggs and that technique is auditable. Employing an emergency threshold, such as 50 eggs, could be established.
Residue determination – points out guidance in industry.
Baird: How might fenbendazole residues impact health issues, especially when eggs are used for vaccines and cancer treatment? Also seeing some data re: problems with hookworms and potential to grow in the brain. How do we address these concerns?
A: Any vaccines are produced in pathogen free eggs and strictly regulated. Worms would be very highly controlled… doesn’t believe this is a problem, based on the TR. Hookworms are not a parasite of poultry; not aware of problems outside GI tracts and reproductive tracts.
Oakley: Are the birds in the slides organic?
A: Two pictures are not organic.
Oakley: How many square feet were allotted for the organic birds pictured?
A: Doesn’t know
Cornucopia note: Cornucopia is a strong proponent of proper outdoor access for all livestock and poultry. This means that not only the majority of an animal’s life should be spent outdoors, but that that outdoor space is managed to protect the environment and maximize the natural behavior of the livestock in question. Parasites are often a hallmark of overcrowding and poor management in poultry. Farmers who allow their birds appropriate space for the climate and land in question and ensure birds have fresh, non-denuded ground to roam rarely have such issues with parasites.
Alice Runde, National Organic Coalition – Coalition Manager
Strengthening organic enforcement rulemaking is a priority. We should not delay origin of livestock and organic enforcement rules. These rules are critical for businesses and maintaining integrity. We need to address inconsistencies between certifiers as well. NOC requests review of peer review audits, asks NOP to explain its risk-based approach and identify gaps that require further action.
Access is not equal; organic food cannot be accessed by some racial and economic groups. This is a problem. Organic needs to prioritize racial justice. Time and resources need to be dedicated to diverse farmer support, expand work and resources to further justice issues.
Ela: We would like to see NOC and other stakeholders apply to NOSB membership. We need diverse members to apply.
Oakley: I have witnessed barriers to certified smaller–scale farmers when language is a barrier—it would greatly increase the number of producers, especially small farmers, that would become certified organic.
Romero-Briones: It is critical for our community to find ways to be more inclusive. Appreciate your thoughts and hope it is reflected in NOSB makeup.
Garth Kahl, Common Treasury Farm (small diversified crop and livestock operation), Independent Organic Services, Inc. (inspection and consulting company)
Board has written comments. Not going to add to those except to say it’s easy to limit liquid fish products not from waste.
Tells story of a processing facility and commitment to staying open, illustrating that it is a good time during the crisis to bring new customers to the local food movement. These potential changes could come out of the COVID-19 crisis. Processors will face significant hardships given the COVID- 19 crisis. Please leave them options on the list.
Bjarne Pedersen, Ellepot A/S, Denmark – Consultant
The motion for paper pots is fine, but NOSB should alter the limit for bio-based for no more than 80% at this stage. This will be difficult but possible. Bio–degradability is the most important thing for Ellepot, more important than origin of materials. Recommend that grower be able to use materials in stock for this season. Due to COVID situation, testing in labs has been delayed. Ask that paper pots are accepted for this season given the delay.
Some 100% bio-based substances in the market will not degrade. For example, wood-based may not be useful for grower. 100% bio-based products can hold up for 6-8 weeks. Useful as a mulch cover, but it’s not useful for every purpose.
Ela: If we stuck with 85%, would that eliminate some of your products from being included?
A: It would eliminate one of them, yes.
Ela: How would we make an annotation to keep cellulose-based materials on there? Most are synthetic, based on paper processing itself. How would you word that annotation?
A: Standard for testing bio-degradation of a product exists. 90% degradation over 2 years is the most common standard used. 17556 in ISO standard I believe.
Ela: So if we stick with 80% based and remaining has to be bio-degradable, would that make sense?
A: Yes, that would be a good way to move in the future.
Christie Badger, National Organic Coalition
Inerts: continues to be unconscionable delay in replacing obsolete references to replacing EPA list 3 and 4. NOSB should not delay in evaluating NPE. We outline a process for moving forward. NOC wants inerts reestablished as a working group.
Paper pots, intention of degrading in soil vs. removing: generally supportive of annotation. Strongly encourage Board to move toward biodegradability.
Both bio–based and biodegradability are equally important. Biodegradation must be considered in a broad way. NOC acknowledges bio–based and biodegradation would be great but acknowledge agroeconomic and health. More research is needed.
Fenbendazole: opposed as proposed. While FDA may consider 2.4 ppm, organic is not set up to mimic conventional. Organic follows precautionary principle. Withdrawal period for fenbendazole needs to have same high standards as other forms of production.
Bradman: Would you change bio-based annotation to 100% biobased?
A: Bio–based and biodegradability are equally important. Don’t think just considering biodegradability alone is sufficient.
Bradman: How about other petroleum products?
A: Will talk about it and try to get some thoughts for you on that.
Bradman: A lot of concern about situation with inerts. Agree with some of your concerns and want to fix that.
A: Hope NOP recognizes they have wonderful research in you and hope they will take advantage of that.
Terry Shistar, Beyond Pesticides – Board Member
Our comments followed several topics: Inert ingredients in pesticides, marine materials, contaminated inputs, and products of fermentation
Requested a comprehensive review of sanitizers and disinfectants. Disinfectants are being reviewed by BP due to COVID. BP’s experience may be helpful in reviewing uses for organic production. NOSB review should follow available disinfectants identified by the EPA as 1. Identify needs, 2. Identify available materials, and then 3. Review according to OFPA criteria.
Emily Brown Rosen, Organic Research Associates
Was an NOP staff member and am glad to be facilitator for inerts working group. Ultimately developed a proposal that was reflected in 2015 NOP recommendations. This change would provide for collaboration with EPA. Quite a few questions as to what the wording means in proposed new annotation. NOSB did side by side comparison of OFPA and EPA regulations and working group thought criteria could be reviewed. We found EPA to be interested in collaborating with inerts working group.
Oakley: What are the consequences of NOSB recommendations to list?
A: No terribly good answers. (1) Do nothing which is where we stand right now; (2) to allow all inerts that have a tolerance exemption by EPA (go along with EPA, which would be more coherent with other agencies and countries), or (3) could fall back on 2012 recommendation to review inerts in products singly or in groups (would be petitions and reviews every five years). Trying to work with EPA would be best way forward.
Michael Sligh, Global Alliance for Organic Integrity
Working to strengthen organic assurance worldwide, working to develop better tools for preventing fraud.
- Remote surveillance, urge cooperation and collaboration to ensure consistent oversight, need potential warning of hotspots for fraud.
- Need to shift greater focus on risk, consistent criteria for risk assessment. Histories of non-compliance, complex chains, volatile areas, high demand commodities, scale of impact if fraud is found, etc. indicate high risk.
- Critical proposed organic import rule get out for public comment ASAP. We don’t want to see rule come out partially and get caught in a change in administration.
- Support calls for supporting racial equity in organic.
Cynthia Fabian, Organic products to eradicate harmful pests in the agricultural market.
Has a patented product that uses gamma rays and in process of getting organic certification. It’s highly effective in killing worms. I’m working on eradicating screw worms. I need to get it to market.
A: Invited to work with NOP staff.
Laura Batcha (unconfirmed), Organic Trade Association
OTA’s strongest asset is diversity and breadth of members. Half of OTA members are small businesses/small farmers reporting less than $1 million in annual sales.
Comments submitted are on behalf of our membership, to help NOSB understand the effect on a broad array of the marketplace. Conducted surveys on a number of topics to inform their written comments.
Mark Keating, Wheel of Life Consulting
Between 1999 and 2002, was a lead specialist at AMS. Fenbendazole: need to include turkeys in the conversation. Worms are a recurring phenomenon in all poultry because increasing outdoor access increases exposure. Farmers and companies petitioned have voluntarily complied with standards. Unethical to leave birds outdoors without this. Does not agree with NOC comments. We have registered pesticides that have tolerances; this is no different. Issue has been poorly managed. “Consumer expectations” is a false foundation for setting standards.
Baird: Had not considered turkeys, but agrees that producers I see having issues with worms are ones that are voluntarily adhering to advanced livestock proposal–and they are seeing a lot of worms in their eggs.
A: Sometimes organic poultry sectors gets painted with broad brush. There is large scale with humane compliance and large scale without.
Baird: People I deal with are small and reporting a lot of worms in eggs.
Bradman: You acknowledge EPA tolerances. Do you have opinion on withdrawal?
A: Does not favor withdrawal period. From compliance standpoint, withdrawal would be very complicated. Does not see reason for standard withdrawal period.
Amber Pool, CCOF
Cancelled
John Foster, 1) EarthKind 2) That Upstart Crow 3) Carbon Catalytics – Director of Business Development, used to be on NOSB (handler)
EPA list 4 concern on inerts: these inerts are essential for organic. They should be continued to be included, as written.
EarthKind: manufacture repellents, use agriculture oils and essential oils to repel pests (primarily for structural uses). Development of better products is stifled. These products lessen the need for synthetic poisons. 2015 recommendation should be implemented. Individual sunset review of every inert is untenable.
Q: How would using the Safer Choice Program affect business environment?
A: It would provide clarity and consistency; allows businesses to bring things to market with some certainty they can be used.
Bradman: What are your comments on question 4?
A: Without an alternative, a number of tools would leave the market—handlers refer to 272/271 through National List even though inerts are not listed per se for handlers. As tools leave the market, without alternatives and clarity on what to develop next, there is little incentive to develop new things. Organic is a small portion of what chemical manufacturers make, so lack of clarity is going to be harmful for development.
Shannon McCormick, Oregon Tilth, Inc.
Not present; skipped
Tom “Geo” Honigford, Hurricane Flats Farm
Farmer calling from his field. Got a kick out of OTA suggesting small farmers report $1 million in sales in a year.
Paper pots transplanter is a critical tool. Plants faster and is cheaper, allowing him to use less labor.
Seen a growing use of plastics. At a big conference in NH there must have been six workshops on how to use plastics in the field. We need to start a conversation about how we can reduce use of plastic and not how we can increase it.
Oakley: Are you advocating for biodegradeable/bio–based mulch?
A: Not advocating for anything. Just expressing concern about plastics and the need to start a conversation.
Q: Do you use paper pots in field? How many years?
A: 8–10 years
Q: Impacts to soil?
A: None. Can see remainder of chains from last years. Nothing from two years ago. It’s gone within a season.
Maricela Adrian, JFDC
Skipped; not present
Beth Rota, Quality Certification Services
Hopes that future meeting will be held in person.
QCS supports adding paper-based crop planting aids to the National List. The proposal is concerning due to its complexity. It should allow anything that decomposes into the soil. Maximum of 15% synthetic fibers is supported, for example, sounds good, but it should be confirmed this matches what farmers currently need.
Supports increasing bio-based fiber content over time. Commercial availability requirement. A simplified National List motion should be passed by NOSB regarding paper planting aids.
Diana Castillo
Skipped; not present
Carlos Chinchilla, Aceites S.A.
Skipped; not present
Keith Jones, Biological Pesticide Industry Alliance – Executive Director
Non-profit, dedicated to responsible development of safe and biological products like bio-pesticides, fertilizers, etc. BPIA has many member companies, many producing products specifically for organic growers.
3011—Procedure National List guidelines document: giving up confidential information just to have something listed; defining “essential” as needed; need to include “best case” timeline. Request to permit inert ingredients with EPA exception for no limits to be allowed without limits.
Cynthia Smith, Biological Pesticide Industry Alliance – Member
Pesticides registration consultant. Past NOP petitioner, currently working on petition.
Inert ingredients: law for organic foods was passed 20 years ago, and there has been much pesticide development since that time. If the rules were to change now, we would use 20 years of development. Companies would lose their investment; clearly some products would be lost. There would be a lot of testing needed for inert ingredients; it would take multiple years through efficacy requirements. It would be preferable to expand the definition to include all inerts that have a tolerance exemption because they do comply with EPA saying they do no harm. Encourage competition within organic market. This would give farmers more access to beneficial inerts that are locked out of organic marketplace. Businesses may not make decision to pursue organic product development.
Ela: We know that list 4 is outdated and cannot be updated at this point. Would you be comfortable if we migrated to safer choice program?
A: If you eliminate list 4 and go to another program it would be a disservice. Preferable to offer an additional program that would offer chances for new products.
Bradman: There may be some materials on list 4 that do not meet OFPA requirements and may not be acceptable for organic. So there is tension there; materials would still sometimes be lost. As long as the materials that are maintained are compatible with organic, then maybe we don’t have a problem with that. There has to be some process to streamline that.
A: There would need to be a long phase-in period for transition (10 years) if you remove anything. Phase in for any new program.
Lee Frankel, Coalition for Sustainable Organics (hydroponic and container trade association) – Executive Director
Climate change will disrupt crop production. Growers use 90% less water using containers. Disagrees with lawsuit that seeks to get rid of container systems in organics.
Jenna Pugliese, NativeEnergy, Inc. – Director of Programs
NativeEnergy is involved in carbon outfit project development to reduce greenhouse gas production on farms. Bio-char is currently prohibited. We maintain biochar is not ash and should be allowed. It can help with carbon sequestration and has soil building qualities.
Ideally all dairy would be pasture raised, but until then, agriculture will be a significant contributor to greenhouse gases.
Wood: What is your perspective regarding hydrolyzing manure?
A: Will get back to you.
Ela: To clarify, bio char is not ash?
A: Correct. Biochar is not ash.
Adam Seitz, Quality Assurance International – Policy Specialist
Certifiers performing on-site inspections is a problem with COVID. QAI asks that an emergency allowance of virtual inspections be allowed.
Malic acid: support re-classification as an allowed synthetic. If the substrate is directly acted on to produce the product in question, it should dictate the status of the final product; if it just feeds the ferment, then it does not affect the final status.
Comment on ion exchange.
NOSB members: Work is being done to keep inspection process moving forward during COVID.
Oakley: Virtual inspection might have a role to play now during COVID, but any virtual inspection now would need to be followed up with an in-person inspection later. That initial inspection is especially important for new producers.
Romero-Briones: Comment on WIC: It’s important to break down institutionalized socio-economic barriers for purchasing in these programs. Will follow up with this issue.
A: it would be great to subsidize and get those organic products to people who need them.
Tucker: Comment on inspections: The USDA organic regulations drive compliance; encouraging critical thinking on ways to ensure compliance in these times, but we’ve gotten questions about on–site inspections. Virtual inspections cannot replace on-site inspections. This is necessary for organic integrity. At this time other methods can be used until an on-site inspection can be performed. This is what we have shared with certifiers.
Oakley: Thank you, Dr. Tucker, for your clarification and reassuring words.
Jill Smith, Western Organic Dairy Producers Alliance, dairy producer at Pure Éire Dairy
WODPA represents approximately 285 dairies across the US. Smith is also an organic dairy producer in WA state.
Supports removal of whey protein from National List. Looks forward to final rule on Origin of Livestock; credibility of organic milk depends on it. Need strict adherence to standards. Smaller scale producers have had to navigate ups and downs of market and it is essential we support producers by leveling the playing field. Also supports addressing climate change. Organic standards encourage consumers to vote with dollars. Must support organic integrity and support organic farms.
Jessica Shade, The Organic Center – Director of Science Programs
Current projects of the Organic Center, informed by current NOSB research priorities:
- Plant disease and insect management—recently got some federal funding to approach this issue. Applying for full OREI funding in the next proposal
- Pathogen prevention—there is some misinformation that there is some risk of introduction of human pathogens.
- Climate change—working on several projects related to climate change mitigation.
Suggestions for research:
- Soil health
- Protection for organic farmers from pesticide residues
- Comparison of pesticide, hormone, and antibiotic residues between organic and conventional products
- Celery powder research should also be included in this year’s research priorities.
Megan DeBates, Organic Trade Association
OTA is committed to protecting the NOSB. A large majority of problems are due to a lack of clear standards. NOP has not enacted many NOSB recommendations. Stakeholders have to advance the standards and work toward continuous improvement. Several important recommendations that have not been implemented by NOP. Years have gone by without NOP addressing NOSB recommendations. OTA is working on legislation that would require NOP to implement or explain reasons for not implementing NOSB recommendations.
Johanna Mirenda, Organic Trade Association
Paper-based crop planting aids: we support most of the aspects of the proposal. The NOP has approved use of these materials temporarily in order to give the NOSB time to review them.
List 4 inerts: it’s time to prioritize 2015 NOSB recommendation to update this listing. List 4 is old and out of date. We are excluded from using EPA’s current program, and manufacturers are unable to invest in new products.
Seaweed and fish-based inputs: concern regarding how NOSB is evaluating “environmental impact” for these materials. Different subcommittees are using inconsistent requirements for same material. Encourage NOSB to create new mechanism for coordinating what it means for marine material harvest to be harmful for the environment.
Tucker: We have not approved task force moving forward; there may be better ways to coordinate between subcommittees.
Oakley: It is possible to collaborate, but the technical expertise of a task force would be beneficial; difficult to carve out time in subcommittees during our normal meetings. “Fertility use” is slipping through the cracks.
Doug Currier, The Organic Materials Review Institute (OMRI)
Technical Director at OMRI. Supports L-Malic Acid. [Technical trouble: unable to hear remainder of testimony.]
Jackie DeMinter, MOSA – Certification/Policy Manager
MOSA certifies over 200 laying hen operations and over 500 vegetable operations.
Consider annotation for BDM film—it is illogical to have a listing that is impossible to meet.
Supports listing of paper as a planting aid. Does the listing include things that are not currently in use? Should it include bio-based testing for everything or just the fibers, for example? This needs further discussion. The bio-based content of the fibers is important—please read MOSA’s written comments.
Fenbendazole: we need further clarification of what “emergency” means. We request this guidance for all livestock: emergency needs to be defined. MOSA has received few requests for a parasiticide.
Baird: You note you have few requests for parasiticides, but Missouri farmers report high need. There seems to be a wide difference in opinion.
A: Not receiving requests from clients to use parasiticides; have not heard about parasite load. Likely because farmers know it’s not an allowed input. Client in Missouri said it would be a nice tool for toolbox but agreed it should not be a replacement for good management practices.
Baird: NOSB agrees “emergency” needs to be defined.
Jason Houle, general public
Skipped; not present
Angela Wartes‐Kahl, Independent Organic Services, Inc.
Skipped; not present
Leslie Touzeau, Quality Certification Services (QCS) – Material Review Specialist
Inert ingredients: Supports new system for inert ingredient review. System in use now is obsolete. 2015 NOSB recommendation should be implemented. Reviewing each individual ingredient would be overly burdensome. Inert ingredients are necessary for many pesticide products. The goal of the 2015 NOSB recommendation was to make the transition to a new system as seamless as possible. Need reasonable implementation time. Critical we have a reliable system in place to evaluate inert materials and that they continue to be available to our organic producers.
Dr Alex Strauch, veterinarian (Was not on speaker list)
Fenbendazole works in conventional production, and there is a need in organic for actual emergency situations. It’s an animal health issue, but also a “kitchen table” issue if consumers find worms in their eggs and lose confidence in products. Organic by design puts these birds at a higher risk. Diatomaceous Earth is ineffective; herbs are ineffective. Using fenbendazole for emergency is a logical progression; we do not currently have an effective alternative.


